Edexcel iGCSE Chemistry -2.11 Combustion Reactions- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -2.11 Combustion Reactions- Study Notes- New syllabus
Edexcel iGCSE Chemistry -2.11 Combustion Reactions- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
2.11 describe the combustion of elements in oxygen, including magnesium, hydrogen and sulfur
2.11 Combustion of Elements in Oxygen
Combustion is a chemical reaction in which a substance reacts rapidly with oxygen, releasing heat and usually light.
Combustion reactions are exothermic.
1. Combustion of Magnesium
Magnesium burns in oxygen with a bright white flame.
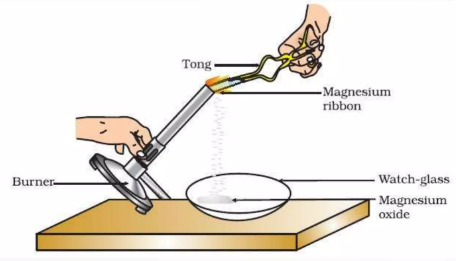
\( \mathrm{2Mg(s) + O_2(g) \rightarrow 2MgO(s)} \)
- Bright white light is produced.
- A white powder (magnesium oxide) forms.
- The reaction is very exothermic.
Magnesium oxide is a basic oxide.
2. Combustion of Hydrogen
Hydrogen burns in oxygen with a pale blue flame.
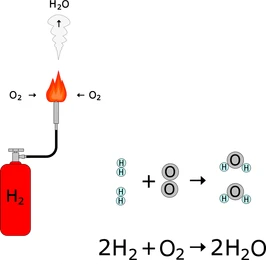
\( \mathrm{2H_2(g) + O_2(g) \rightarrow 2H_2O(l)} \)
- A squeaky pop may be heard if ignited.
- Water is formed.
- The reaction releases heat energy.
This reaction is often used to test for hydrogen gas.
3. Combustion of Sulfur
Sulfur burns in oxygen with a blue flame.
\( \mathrm{S(s) + O_2(g) \rightarrow SO_2(g)} \)
- A sharp, choking smell is produced.
- Sulfur dioxide gas forms.
- The reaction is exothermic.
Sulfur dioxide is an acidic oxide.
| Element | Flame Colour | Product Formed | Type of Oxide |
|---|---|---|---|
| Magnesium | Bright white | \( \mathrm{MgO} \) | Basic |
| Hydrogen | Pale blue | \( \mathrm{H_2O} \) | Neutral |
| Sulfur | Blue | \( \mathrm{SO_2} \) | Acidic |
Example 1 (Conceptual):
Why is combustion described as an exothermic reaction?
▶️ Answer/Explanation
Energy is released as heat and often light.
The surroundings become hotter.
Example 2 (Application):
Write the balanced equation for the combustion of magnesium.
▶️ Answer/Explanation
\( \mathrm{2Mg(s) + O_2(g) \rightarrow 2MgO(s)} \)
Example 3 (Hard ):
Explain fully how the combustion of magnesium, hydrogen and sulfur demonstrates the formation of different types of oxides.
▶️ Answer/Explanation
All three elements react with oxygen to form oxides.
Magnesium forms magnesium oxide, which is a basic oxide.
Hydrogen forms water, which is neutral.
Sulfur forms sulfur dioxide, which is an acidic oxide.
This shows that metals tend to form basic oxides, while non-metals form acidic oxides.
