Edexcel iGCSE Chemistry -2.12 Formation of Carbon Dioxide- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -2.12 Formation of Carbon Dioxide- Study Notes- New syllabus
Edexcel iGCSE Chemistry -2.12 Formation of Carbon Dioxide- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
2.12 describe the formation of carbon dioxide from the thermal decomposition of metal carbonates, including copper(II) carbonate
2.12 Formation of Carbon Dioxide from the Thermal Decomposition of Metal Carbonates
Thermal decomposition is a reaction in which a compound breaks down into simpler substances when heated.
Many metal carbonates decompose when heated to produce:
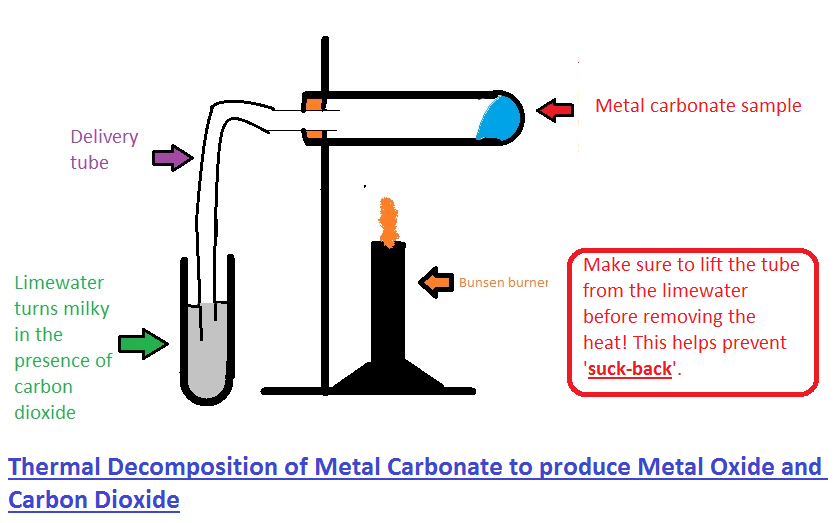
- A metal oxide
- Carbon dioxide gas
General Equation for Metal Carbonates
\( \mathrm{MCO_3(s) \rightarrow MO(s) + CO_2(g)} \)
Where \( \mathrm{M} \) represents a metal.
Example: Copper(II) Carbonate
Copper(II) carbonate is a green solid.
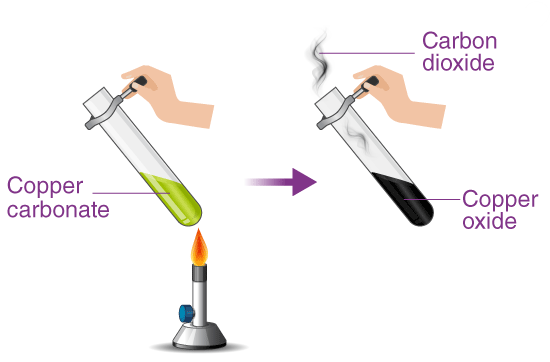
\( \mathrm{CuCO_3(s) \rightarrow CuO(s) + CO_2(g)} \)
- Green solid turns black.
- Black copper(II) oxide is formed.
- Carbon dioxide gas is released.
Testing for Carbon Dioxide
Carbon dioxide can be confirmed by bubbling the gas through limewater.
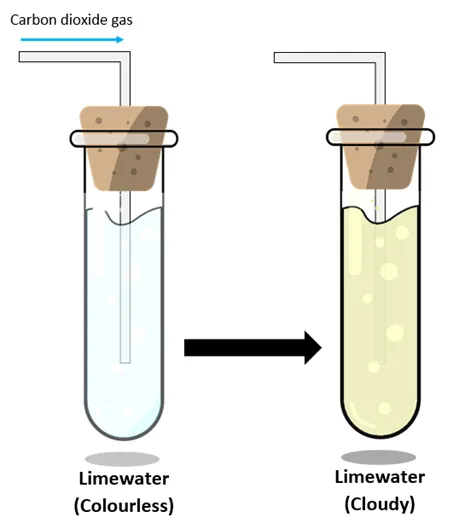
\( \mathrm{Ca(OH)_2(aq) + CO_2(g) \rightarrow CaCO_3(s) + H_2O(l)} \)
- Limewater turns milky (cloudy).
- This confirms the presence of carbon dioxide.
Energy Change
- Thermal decomposition requires heat.
- It is an endothermic reaction.
| Metal Carbonate | Colour Before Heating | Metal Oxide Formed | Gas Produced |
|---|---|---|---|
| \( \mathrm{CuCO_3} \) | Green | Black \( \mathrm{CuO} \) | \( \mathrm{CO_2} \) |
| \( \mathrm{CaCO_3} \) | White | White \( \mathrm{CaO} \) | \( \mathrm{CO_2} \) |
Example 1 (Conceptual):
Why is thermal decomposition described as endothermic?
▶️ Answer/Explanation
Heat energy is required to break bonds.
Energy is taken in from the surroundings.
Example 2 (Application):
Write the balanced equation for the thermal decomposition of calcium carbonate.
▶️ Answer/Explanation
\( \mathrm{CaCO_3(s) \rightarrow CaO(s) + CO_2(g)} \)
Example 3 (Hard ):
Describe fully what is observed and what happens when copper(II) carbonate is heated strongly.
▶️ Answer/Explanation
The green copper(II) carbonate is heated.
It decomposes because thermal energy breaks chemical bonds.
The green solid turns black as copper(II) oxide forms.
Carbon dioxide gas is produced.
The gas can be tested using limewater, which turns milky.
The reaction is endothermic because heat is absorbed.
