Edexcel iGCSE Chemistry -2.14 Practical: Oxygen in Air- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -2.14 Practical: Oxygen in Air- Study Notes- New syllabus
Edexcel iGCSE Chemistry -2.14 Practical: Oxygen in Air- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
2.14 practical: determine the approximate percentage by volume of oxygen in air using a metal or a non-metal
2.14 Practical: Determining the Approximate Percentage by Volume of Oxygen in Air
This practical determines the percentage of oxygen in air by removing oxygen using either:
- A metal (e.g. iron)
- A non-metal (e.g. phosphorus)
The decrease in gas volume represents the volume of oxygen present.
Method 1: Using Iron (Rusting Method)
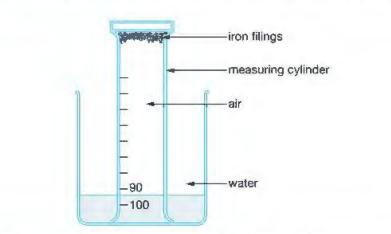
- Damp iron wool is placed inside a measuring cylinder.
- The cylinder is inverted over water in a trough.
- The initial volume of trapped air is recorded.
- The iron reacts slowly with oxygen to form iron oxide (rust).
- Water rises inside the cylinder.
- The final volume of gas is recorded after several days.
Reaction:
\( \mathrm{4Fe(s) + 3O_2(g) \rightarrow 2Fe_2O_3(s)} \)
Method 2: Using Phosphorus (Combustion Method)
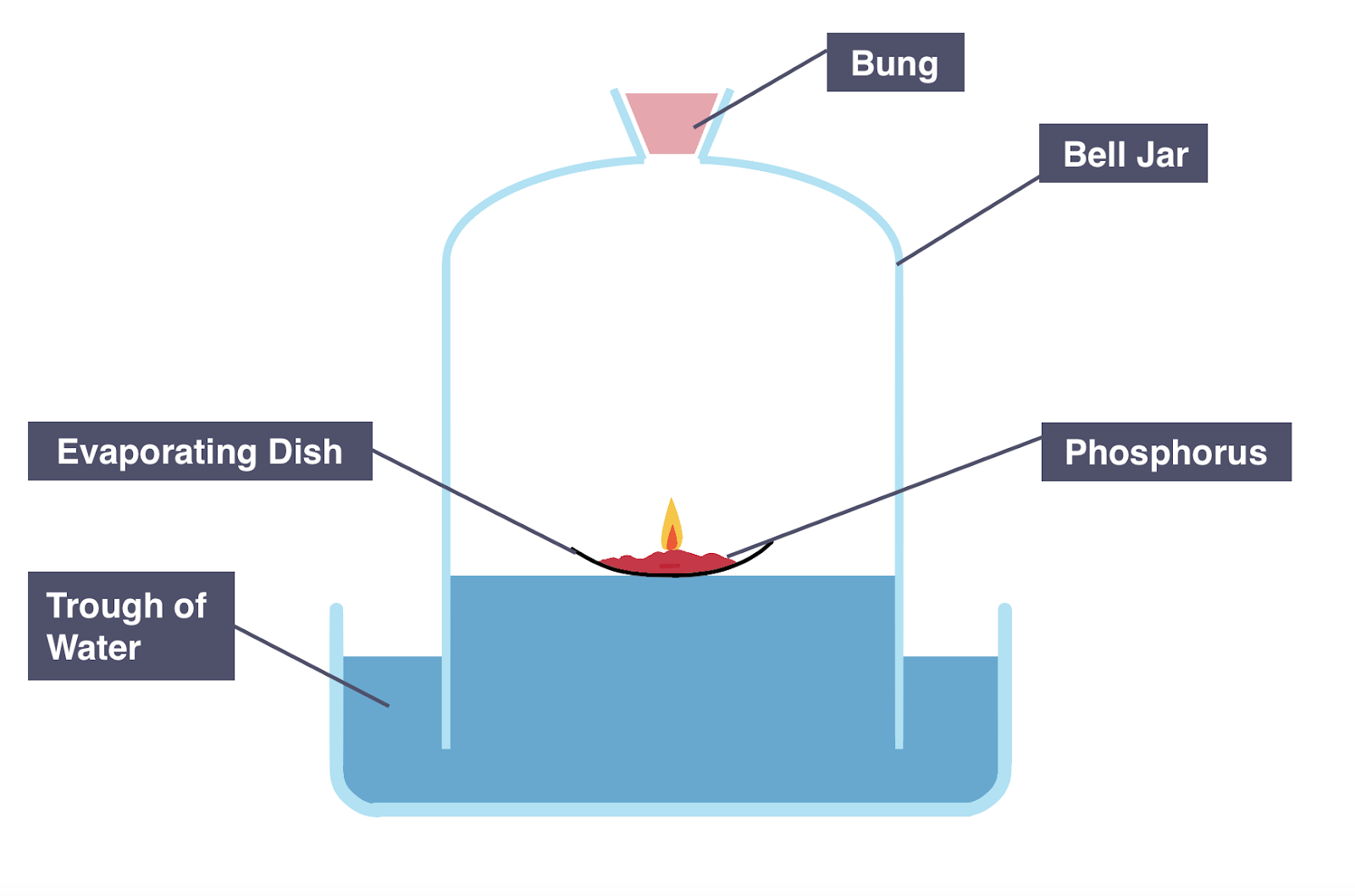
- A small piece of phosphorus is placed on a heatproof dish.
- It is ignited inside a sealed container over water.
- Phosphorus reacts rapidly with oxygen.
- White smoke of phosphorus(V) oxide forms.
- Water rises as oxygen is removed.
- Initial and final volumes are measured.
Reaction:
\( \mathrm{4P(s) + 5O_2(g) \rightarrow P_4O_{10}(s)} \)
Calculation of Percentage Oxygen
\( \mathrm{Percentage\ of\ oxygen = \dfrac{Volume\ decrease}{Initial\ volume} \times 100} \)
The volume decrease corresponds to the oxygen that reacted.
Expected Result
- The volume decreases by approximately one-fifth.
- This shows oxygen makes up about 20–21% of air.
| Stage | What Happens | Why |
|---|---|---|
| Initial setup | Known volume of air trapped | Starting measurement |
| Reaction occurs | Oxygen reacts with metal/non-metal | Oxygen removed from air |
| Water rises | Gas volume decreases | Space left by oxygen |
| Final measurement | New volume recorded | Used for calculation |
Example 1 (Conceptual):
Why must the container be sealed during the experiment?
▶️ Answer/Explanation
To prevent air from entering or escaping.
This ensures only oxygen reacts.
Accurate volume measurements can be made.
Example 2 (Numerical):
An experiment begins with 80 cm³ of air. After reaction, 64 cm³ remains. Calculate the percentage of oxygen in air.
▶️ Answer/Explanation
Volume decrease = \( \mathrm{80 – 64 = 16\ cm^3} \)
\( \mathrm{Percentage = \dfrac{16}{80} \times 100 = 20\%} \)
Example 3 (Hard):
Explain fully how the experiment using phosphorus determines the percentage of oxygen in air and why the result is only approximate.
▶️ Answer/Explanation
Phosphorus reacts completely with oxygen inside a sealed container.
This removes oxygen from the trapped air.
The gas volume decreases as oxygen is used up.
Water rises to replace the lost volume.
The decrease in volume represents the volume of oxygen in the air.
Dividing this by the initial volume and multiplying by 100 gives the percentage.
The result is approximate because small experimental errors may occur.
Some oxygen may remain unreacted.
There may be slight measurement inaccuracies.
