Edexcel iGCSE Chemistry -2.15 Reactivity Series from Reactions with Water and Acids- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -2.15 Reactivity Series from Reactions with Water and Acids- Study Notes- New syllabus
Edexcel iGCSE Chemistry -2.15 Reactivity Series from Reactions with Water and Acids- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
2.15 understand how metals can be arranged in a reactivity series based on their reactions with:
• water
• dilute hydrochloric or sulfuric acid
2.15 Arranging Metals in a Reactivity Series Using Reactions with Water and Dilute Acids
Metals can be arranged in order of their reactivity based on how easily they react.
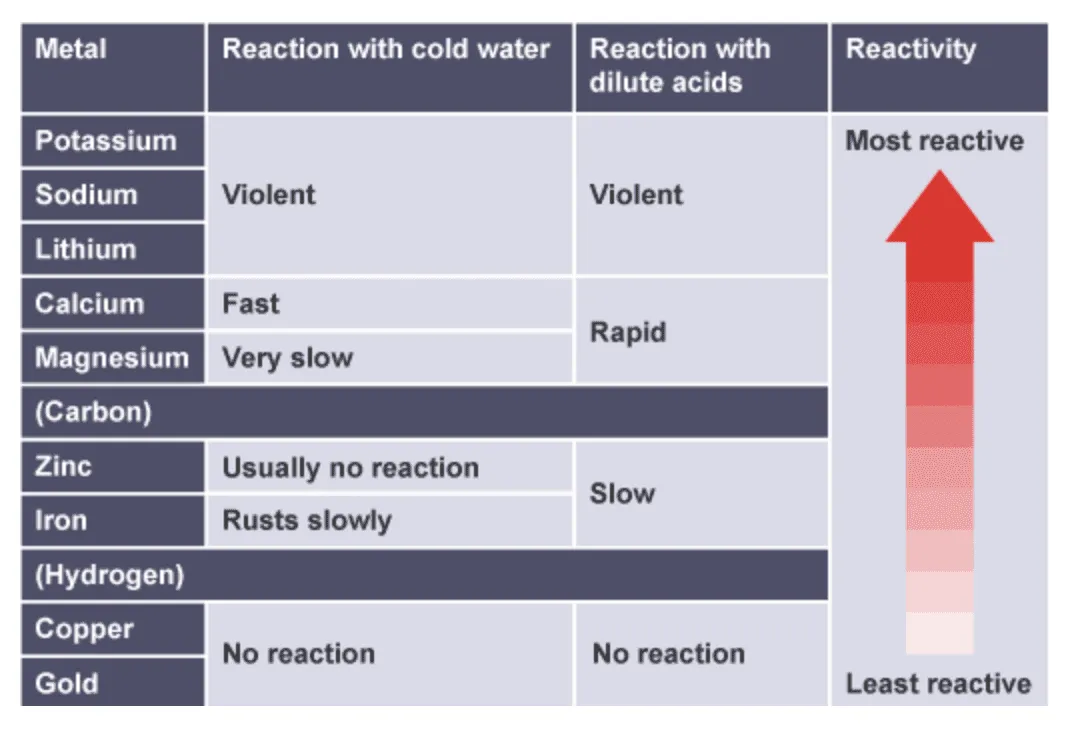
The more reactive the metal, the more easily it loses electrons to form positive ions.
\( \mathrm{M \rightarrow M^{n+} + ne^-} \)
1. Reactions with Water
Very reactive metals react with cold water.
General equation:
\( \mathrm{2M(s) + 2H_2O(l) \rightarrow 2MOH(aq) + H_2(g)} \)
- Potassium – very vigorous
- Sodium – vigorous
- Lithium – less vigorous
- Calcium – reacts steadily
- Magnesium – very slow with cold water
- Zinc, iron, copper – no reaction with cold water
More reactive metals react faster and more violently.
2. Reactions with Dilute Hydrochloric or Sulfuric Acid
Metals above hydrogen in the reactivity series react with dilute acids.
General equation:
\( \mathrm{Metal + Acid \rightarrow Salt + Hydrogen} \)
Example (magnesium and hydrochloric acid):
\( \mathrm{Mg(s) + 2HCl(aq) \rightarrow MgCl_2(aq) + H_2(g)} \)
- Magnesium – vigorous reaction
- Zinc – moderate reaction
- Iron – slow reaction
- Copper – no reaction
The speed of hydrogen gas production indicates reactivity.
Resulting Reactivity Series (Partial)
| More Reactive | Reacts with Water? | Reacts with Dilute Acid? |
|---|---|---|
| Potassium | Yes (vigorous) | Yes |
| Sodium | Yes | Yes |
| Magnesium | Very slow | Yes (vigorous) |
| Zinc | No | Yes (moderate) |
| Iron | No | Yes (slow) |
| Copper | No | No |
Metals that do not react with dilute acids are less reactive than hydrogen.
Example 1 (Conceptual):
Why does copper not react with dilute hydrochloric acid?
▶️ Answer/Explanation
Copper is below hydrogen in the reactivity series.
It cannot displace hydrogen from acid.
Example 2 (Application):
If a metal reacts slowly with dilute acid but does not react with cold water, where would it be placed in the reactivity series?
▶️ Answer/Explanation
Above hydrogen (because it reacts with acid).
Below metals like sodium and magnesium (because it does not react with cold water).
Example 3 (Hard):
Explain fully how reactions with water and dilute acids allow metals to be arranged in a reactivity series.
▶️ Answer/Explanation
More reactive metals react vigorously with water to produce a metal hydroxide and hydrogen gas.
Less reactive metals do not react with cold water.
Metals above hydrogen react with dilute acids to produce a salt and hydrogen gas.
The speed and vigour of hydrogen production indicates relative reactivity.
Metals that do not react with acids are below hydrogen.
By comparing these reactions, metals can be arranged in order of decreasing reactivity.
