Edexcel iGCSE Chemistry -2.16 Reactivity Series from Displacement Reactions- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -2.16 Reactivity Series from Displacement Reactions- Study Notes- New syllabus
Edexcel iGCSE Chemistry -2.16 Reactivity Series from Displacement Reactions- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
2.16 understand how metals can be arranged in a reactivity series based on their displacement reactions between:
• metals and metal oxides
• metals and aqueous solutions of metal salts
2.16 Arranging Metals in a Reactivity Series Using Displacement Reactions
Metals can also be arranged in a reactivity series by observing displacement reactions.
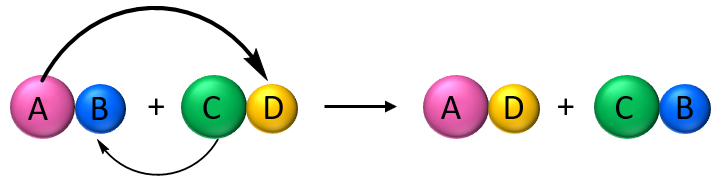
A more reactive metal will displace a less reactive metal from:
- A metal oxide
- An aqueous solution of a metal salt
1. Metal + Metal Oxide Reactions
A more reactive metal can remove oxygen from the oxide of a less reactive metal.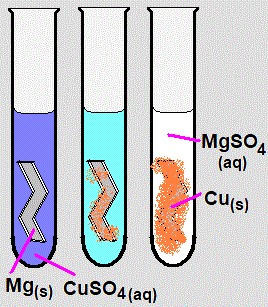
Example: Magnesium and copper(II) oxide
\( \mathrm{Mg(s) + CuO(s) \rightarrow MgO(s) + Cu(s)} \)
- Magnesium removes oxygen from copper oxide.
- Magnesium is more reactive than copper.
- Copper is displaced.
However:
\( \mathrm{Cu(s) + MgO(s) \rightarrow No\ reaction} \)
Copper cannot remove oxygen from magnesium oxide because it is less reactive.
2. Metal + Aqueous Metal Salt Reactions
A more reactive metal can displace a less reactive metal from its salt solution.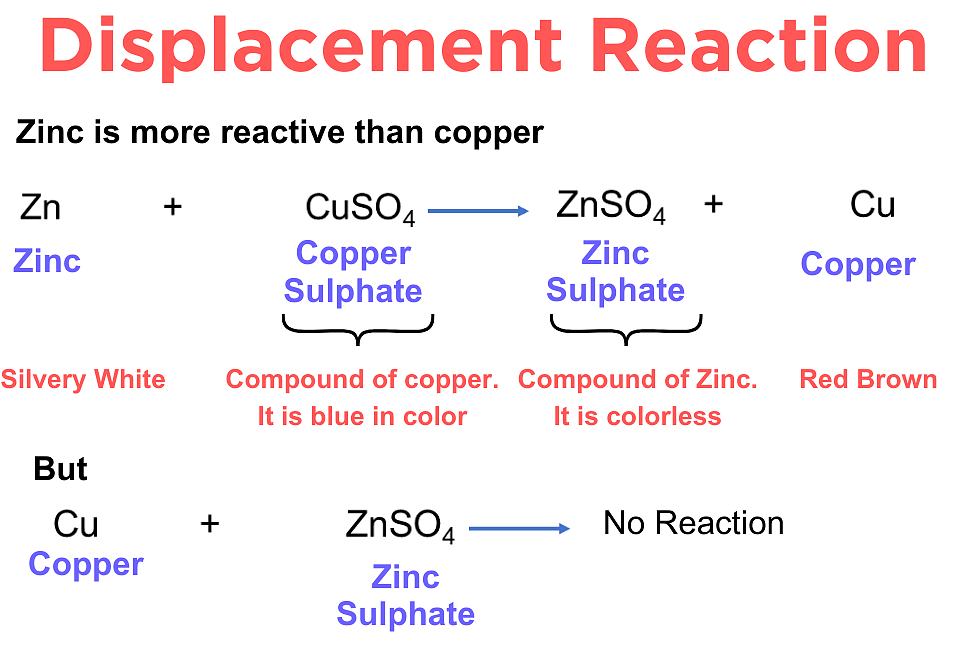
Example: Zinc and copper(II) sulfate
\( \mathrm{Zn(s) + CuSO_4(aq) \rightarrow ZnSO_4(aq) + Cu(s)} \)
- Blue solution fades.
- Brown copper is deposited.
- Zinc is more reactive than copper.
If copper is added to zinc sulfate:
\( \mathrm{Cu(s) + ZnSO_4(aq) \rightarrow No\ reaction} \)
Copper is less reactive than zinc.
What Displacement Shows
- If displacement occurs → metal added is more reactive.
- If no reaction occurs → metal added is less reactive.
- The more reactions a metal can perform, the higher it is in the series.
| Metal Added | Substance | Reaction? | Conclusion |
|---|---|---|---|
| Magnesium | \( \mathrm{CuO} \) | Yes | Mg more reactive than Cu |
| Zinc | \( \mathrm{CuSO_4} \) | Yes | Zn more reactive than Cu |
| Copper | \( \mathrm{ZnSO_4} \) | No | Cu less reactive than Zn |
Example 1 (Conceptual):
Why does magnesium displace copper from copper(II) oxide?
▶️ Answer/Explanation
Magnesium is more reactive than copper.
It has a greater tendency to form ions.
It removes oxygen from copper oxide.
Example 2 (Application):
Predict what happens when iron is added to copper(II) sulfate solution.
▶️ Answer/Explanation
\( \mathrm{Fe(s) + CuSO_4(aq) \rightarrow FeSO_4(aq) + Cu(s)} \)
Iron displaces copper because iron is more reactive.
Example 3 (Hard):
Explain fully how displacement reactions can be used to arrange metals in a reactivity series.
▶️ Answer/Explanation
A more reactive metal will displace a less reactive metal from its oxide or salt solution.
If a metal removes oxygen from another metal oxide, it is more reactive.
If a metal displaces another from a salt solution, it is more reactive.
If no reaction occurs, the metal is less reactive.
By comparing which metals can displace others, the metals can be arranged in order of decreasing reactivity.
