Edexcel iGCSE Chemistry -2.18 Rusting of Iron- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -2.18 Rusting of Iron- Study Notes- New syllabus
Edexcel iGCSE Chemistry -2.18 Rusting of Iron- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
2.18 know the conditions under which iron rusts
2.18 Conditions Required for Iron to Rust
Rusting is the corrosion of iron.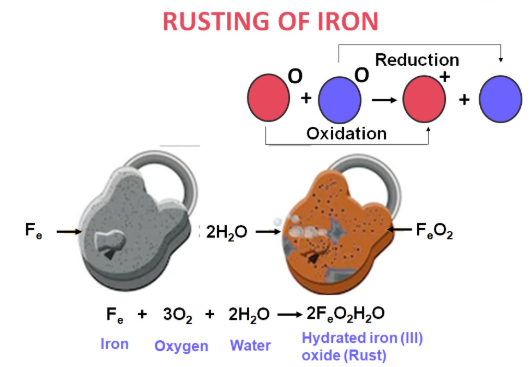
Rust is hydrated iron(III) oxide.
\( \mathrm{Fe_2O_3 \cdot xH_2O} \)
Conditions Needed for Rusting
- Oxygen (from air)
- Water (moisture)
Both oxygen and water must be present for rusting to occur.
Overall Reaction (Simplified)
\( \mathrm{4Fe(s) + 3O_2(g) + xH_2O(l) \rightarrow 2Fe_2O_3 \cdot xH_2O(s)} \)
Experimental Evidence
Three common test conditions:
- Iron in dry air (oxygen present, no water).
- Iron in boiled water covered with oil (water present, no oxygen).
- Iron exposed to both air and water.
| Condition | Water Present? | Oxygen Present? | Rust Forms? |
|---|---|---|---|
| Dry air | No | Yes | No |
| Boiled water + oil | Yes | No | No |
| Air + water | Yes | Yes | Yes |
Important Points
- Salt water increases the rate of rusting.
- Acidic conditions increase the rate of rusting.
- Rusting is faster in coastal environments.
Example 1 (Conceptual):
Why does iron not rust in dry air?
▶️ Answer/Explanation
Water is not present.
Both oxygen and water are required for rusting.
Example 2 (Application):
Why does iron rust more quickly in salt water than in pure water?
▶️ Answer/Explanation
Salt water acts as an electrolyte.
It allows ions to move more easily.
This increases the rate of corrosion.
Example 3 (Hard ):
Explain fully the conditions required for iron to rust and describe how experiments demonstrate this.
▶️ Answer/Explanation
Rusting requires both oxygen and water.
If iron is placed in dry air, oxygen is present but water is absent, so rust does not form.
If iron is placed in boiled water with oil, water is present but oxygen is excluded, so rust does not form.
When iron is exposed to both air and water, rust forms.
This shows that both oxygen and water are necessary conditions for rusting.
