Edexcel iGCSE Chemistry -2.19 Prevention of Rusting- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -2.19 Prevention of Rusting- Study Notes- New syllabus
Edexcel iGCSE Chemistry -2.19 Prevention of Rusting- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
2.19 understand how the rusting of iron may be prevented by:
• barrier methods
• galvanising
• sacrificial protection
2.19 Preventing the Rusting of Iron
Rusting requires both oxygen and water.
Preventing rusting involves stopping iron from coming into contact with oxygen and water, or protecting it chemically.
1. Barrier Methods
Barrier methods prevent oxygen and water from reaching the iron surface.

- Painting
- Oiling or greasing
- Plastic coating
These methods form a physical layer over the iron.
If the coating is scratched, rusting can begin at the exposed area.
2. Galvanising
Galvanising involves coating iron or steel with zinc.
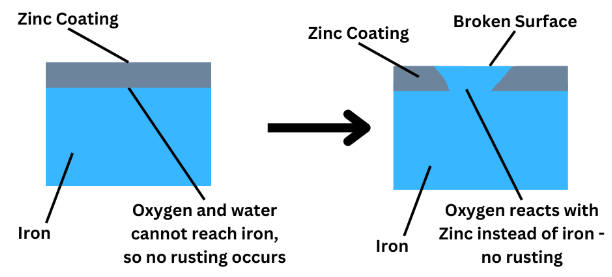
- Zinc forms a protective barrier.
- Zinc is more reactive than iron.
Even if the zinc coating is scratched, zinc will react instead of iron.
\( \mathrm{Zn \rightarrow Zn^{2+} + 2e^-} \)
Zinc loses electrons in preference to iron.
3. Sacrificial Protection
Sacrificial protection uses a more reactive metal attached to iron.
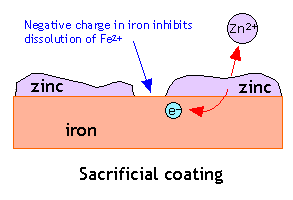
- Common metals used: zinc or magnesium.
- The more reactive metal corrodes instead of iron.
The sacrificial metal loses electrons instead of iron.
\( \mathrm{Mg \rightarrow Mg^{2+} + 2e^-} \)
Iron remains protected because it does not lose electrons.
| Method | How It Works | What Happens if Damaged? |
|---|---|---|
| Barrier method | Blocks oxygen and water | Iron rusts at exposed area |
| Galvanising | Zinc coating + sacrificial action | Zinc protects iron |
| Sacrificial protection | More reactive metal corrodes | Iron remains protected |
Example 1 (Conceptual):
Why does painting prevent rusting?
▶️ Answer/Explanation
Paint forms a barrier.
It prevents oxygen and water from reaching iron.
Without oxygen and water, rusting cannot occur.
Example 2 (Application):
Why is zinc used in galvanising instead of copper?
▶️ Answer/Explanation
Zinc is more reactive than iron.
It can act as a sacrificial metal.
Copper is less reactive than iron and would not protect it.
Example 3 (Hard):
Explain fully how galvanising protects iron from rusting, even if the coating is scratched.
▶️ Answer/Explanation
Galvanising coats iron with zinc.
Zinc acts as a barrier to oxygen and water.
Zinc is more reactive than iron.
If the coating is scratched, zinc reacts and loses electrons instead of iron.
This sacrificial action prevents iron from oxidising.
Therefore the iron remains protected.
