Edexcel iGCSE Chemistry -2.2–2.3 Trends in Reactivity of Group 1 , Properties of Alkali Metals- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -2.2–2.3 Trends in Reactivity of Group 1 , Properties of Alkali Metals- Study Notes- New syllabus
Edexcel iGCSE Chemistry -2.2–2.3 Trends in Reactivity of Group 1 , Properties of Alkali Metals- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
2.2 understand how the differences between the reactions of these elements with air and water provide evidence for the trend in reactivity in Group 1
2.3 use knowledge of trends in Group 1 to predict the properties of other alkali metals
2.2 Trends in Reactivity of Group 1 from Reactions with Air and Water
Group 1 metals (alkali metals) show differences in how vigorously they react with air and water.
These differences provide evidence for a trend in reactivity down the group.
Group 1 Elements Considered

- Lithium (Li)
- Sodium (Na)
- Potassium (K)
1. Reactions with Air (Oxygen)
All Group 1 metals react with oxygen in air to form metal oxides.
General equation:
\( \mathrm{4M(s) + O_2(g) \rightarrow 2M_2O(s)} \)
Observed Differences:
- Lithium tarnishes slowly.
- Sodium tarnishes quickly.
- Potassium tarnishes very rapidly.
The speed of reaction increases down the group.
2. Reactions with Water
General equation:
\( \mathrm{2M(s) + 2H_2O(l) \rightarrow 2MOH(aq) + H_2(g)} \)
Observed Differences:
- Lithium reacts gently (slow fizzing).
- Sodium reacts more vigorously (melts into a ball).
- Potassium reacts very vigorously (may ignite with lilac flame).
The reaction becomes more vigorous down the group.
Trend in Reactivity
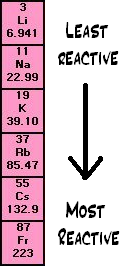
- Reactivity increases down Group 1.
- Potassium is more reactive than sodium.
- Sodium is more reactive than lithium.
Explanation for the Trend
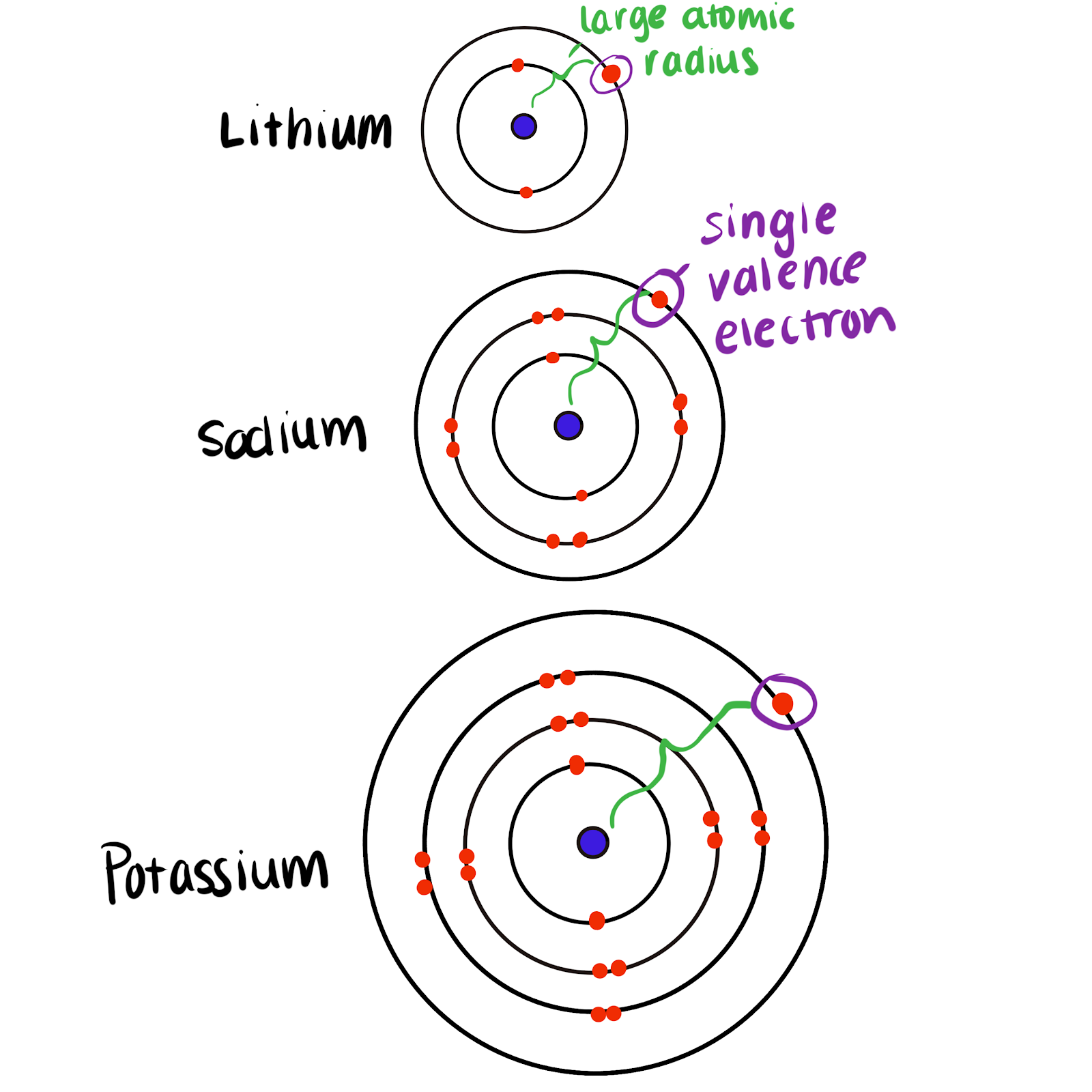
- All Group 1 metals have one outer electron.
- They react by losing this electron.
- Down the group, atoms are larger.
- The outer electron is further from the nucleus.
- The attraction between nucleus and outer electron decreases.
- It is easier to lose the electron.
- Therefore reactivity increases.
| Element | Reaction with Air | Reaction with Water | Relative Reactivity |
|---|---|---|---|
| Lithium | Slow tarnish | Gentle fizzing | Lowest |
| Sodium | Quick tarnish | Vigorous | Medium |
| Potassium | Very rapid tarnish | Very vigorous (may ignite) | Highest |
Example 1 (Conceptual):
Why does potassium react more vigorously with water than lithium?
▶️ Answer/Explanation
Potassium atoms are larger.
The outer electron is further from the nucleus.
The attraction is weaker.
It is lost more easily.
Therefore potassium is more reactive.
Example 2 (Application):
Predict how rubidium (below potassium in Group 1) would react with water.
▶️ Answer/Explanation
It would react even more vigorously than potassium.
Reactivity increases down the group.
Example 3 (Hard):
Explain fully how differences in reactions with air and water provide evidence for the trend in reactivity down Group 1.
▶️ Answer/Explanation
All Group 1 metals react with air and water in similar ways, forming oxides and hydroxides.
However, the speed and vigour of reaction increase down the group.
Lithium reacts slowly, sodium reacts more vigorously, and potassium reacts very vigorously.
This shows that reactivity increases down the group.
Down the group, atomic radius increases and the outer electron is further from the nucleus.
The attraction between nucleus and outer electron decreases.
The outer electron is lost more easily.
This explains the increasing reactivity trend.
2.3 Using Trends in Group 1 to Predict the Properties of Other Alkali Metals
Group 1 elements are known as the alkali metals. They include:
- Lithium (Li)
- Sodium (Na)
- Potassium (K)
- Rubidium (Rb)
- Caesium (Cs)
By understanding the trends down Group 1, we can predict the properties of alkali metals that we may not have directly studied.
Key Trends Down Group 1
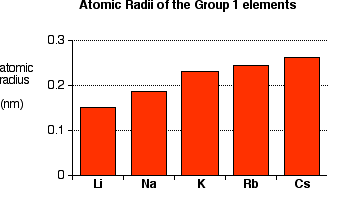
- Reactivity increases.
- Atomic radius increases.
- Melting point decreases.
- Boiling point decreases.
- Density generally increases.
Reactivity Trend
All Group 1 metals have one outer electron.

- Down the group, atoms become larger.
- The outer electron is further from the nucleus.
- The attraction between the nucleus and outer electron decreases.
- The outer electron is lost more easily.
- Reactivity increases.
Therefore, rubidium and caesium will be more reactive than potassium.
Reaction with Water (Prediction)
General equation:
\( \mathrm{2M(s) + 2H_2O(l) \rightarrow 2MOH(aq) + H_2(g)} \)
- Rubidium would react very violently.
- Caesium would react explosively.
- Both would form alkaline hydroxides and hydrogen gas.
Melting Point Trend
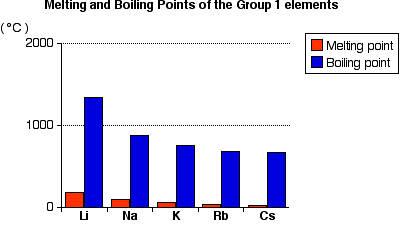
- Melting point decreases down the group.
- Atoms become larger.
- Metallic bonding becomes weaker.
- Less energy is required to melt the metal.
Therefore, caesium has a lower melting point than sodium.
| Property | Trend Down Group | Prediction for Rubidium / Caesium |
|---|---|---|
| Reactivity | Increases | Very high / explosive |
| Melting point | Decreases | Lower than potassium |
| Atomic size | Increases | Larger atoms |
Example 1 (Conceptual):
Why can we predict that caesium is more reactive than sodium?
▶️ Answer/Explanation
Reactivity increases down Group 1.
Caesium has a larger atomic radius.
The outer electron is further from the nucleus.
It is lost more easily.
Example 2 (Application):
Predict what happens when rubidium reacts with water.
▶️ Answer/Explanation
It reacts very violently.
It forms rubidium hydroxide.
Hydrogen gas is produced.
The solution becomes alkaline.
Example 3 (Hard):
Explain fully how knowledge of Group 1 trends allows you to predict the properties of caesium.
▶️ Answer/Explanation
Reactivity increases down the group because atomic radius increases.
The outer electron is further from the nucleus and less strongly attracted.
It is lost more easily, so caesium is extremely reactive.
Melting point decreases down the group due to weaker metallic bonding.
Therefore caesium has a lower melting point than sodium and potassium.
From these trends, we can predict it reacts explosively with water and forms caesium hydroxide and hydrogen gas.
