Edexcel iGCSE Chemistry -2.20 Oxidation and Reduction- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -2.20 Oxidation and Reduction- Study Notes- New syllabus
Edexcel iGCSE Chemistry -2.20 Oxidation and Reduction- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
2.20 understand the terms:
• oxidation
• reduction
• redox
• oxidising agent
• reducing agent
in terms of gain or loss of oxygen and loss or gain of electrons
2.20 Oxidation, Reduction and Redox (Oxygen and Electron Definitions)
Oxidation and reduction are chemical processes that always occur together in a reaction.
Oxidation
Oxidation can be defined in two ways: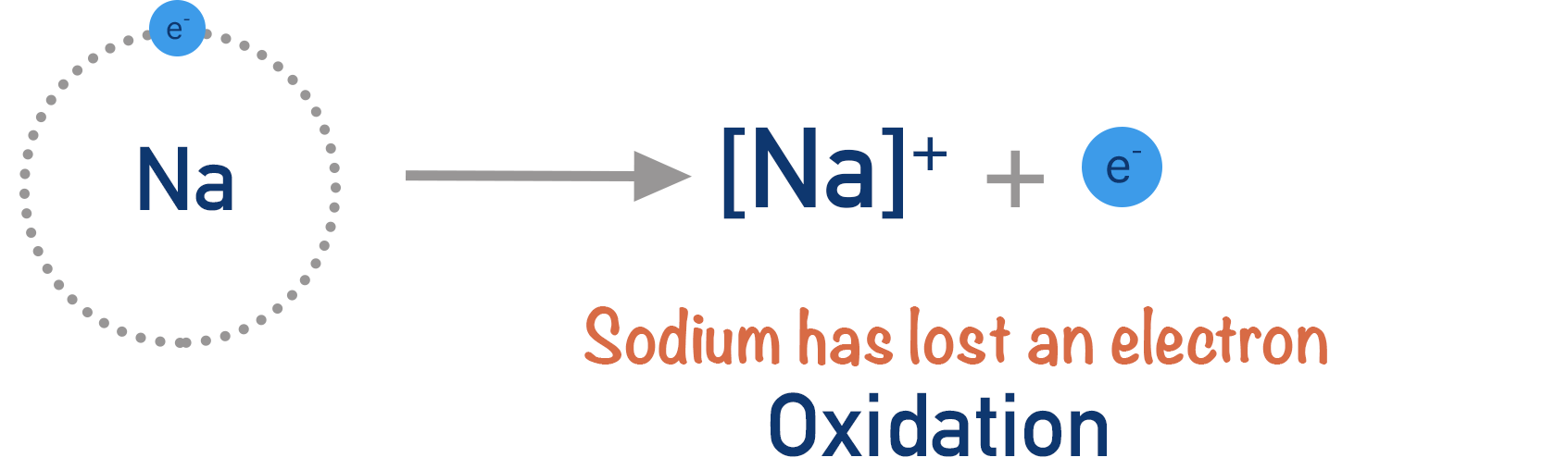
- Gain of oxygen
- Loss of electrons
Example (oxygen definition):
\( \mathrm{2Mg(s) + O_2(g) \rightarrow 2MgO(s)} \)
Magnesium gains oxygen, so magnesium is oxidised.
Example (electron definition):
\( \mathrm{Mg \rightarrow Mg^{2+} + 2e^-} \)
Magnesium loses electrons, so it is oxidised.
Reduction
Reduction can also be defined in two ways: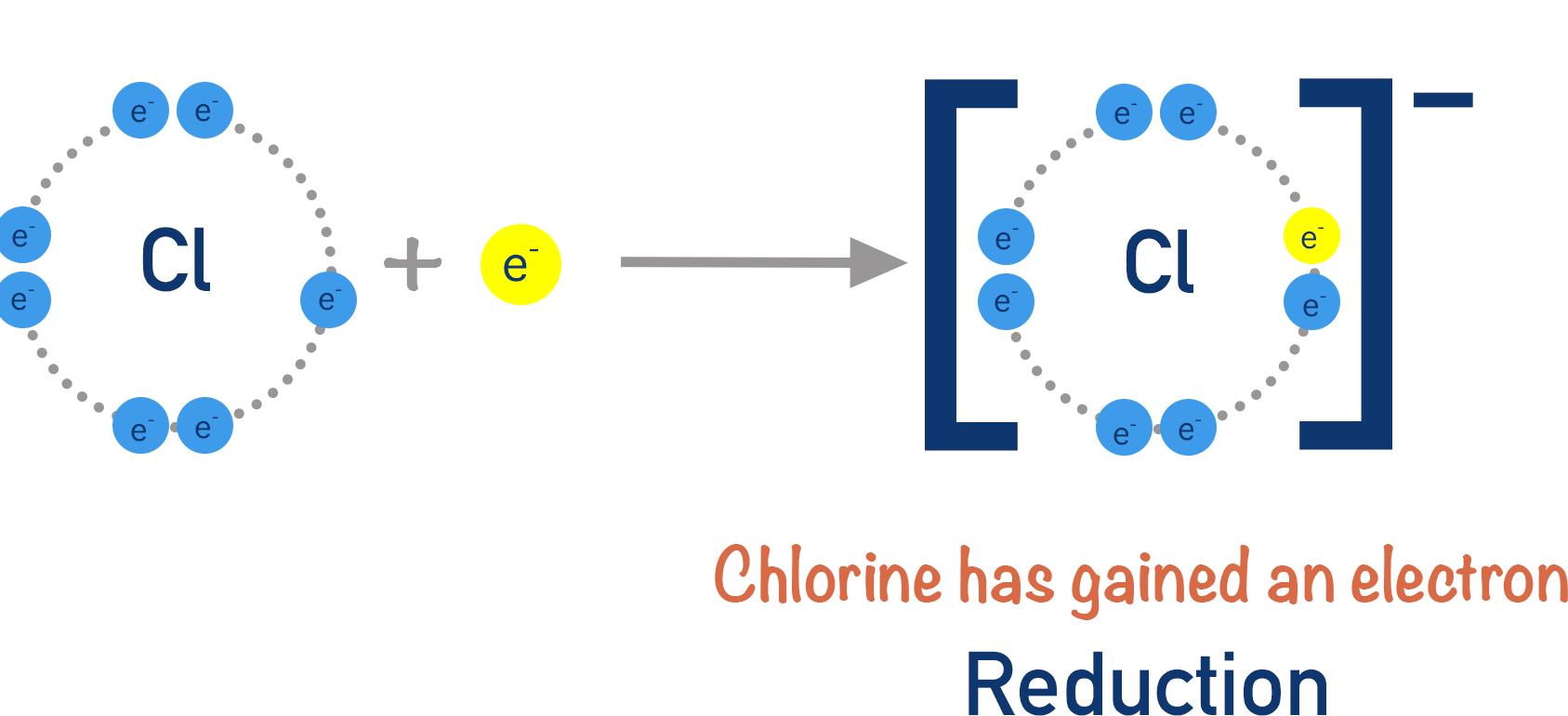
- Loss of oxygen
- Gain of electrons
Example (oxygen definition):
\( \mathrm{CuO(s) + Mg(s) \rightarrow Cu(s) + MgO(s)} \)
Copper(II) oxide loses oxygen, so it is reduced.
Example (electron definition):
\( \mathrm{Cu^{2+} + 2e^- \rightarrow Cu} \)
Copper ions gain electrons, so they are reduced.
Redox Reaction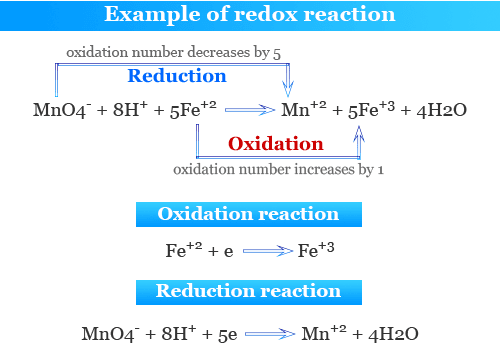
A redox reaction is a reaction in which oxidation and reduction occur at the same time.
In the reaction:
\( \mathrm{CuO + Mg \rightarrow Cu + MgO} \)
- Magnesium is oxidised (gains oxygen / loses electrons).
- Copper(II) oxide is reduced (loses oxygen / gains electrons).
Oxidising Agent
An oxidising agent is a substance that causes another substance to be oxidised.
An oxidising agent: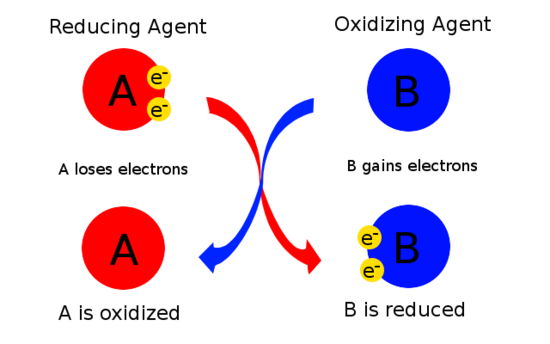
- Gives oxygen to another substance, or
- Gains electrons
It is itself reduced.
Reducing Agent
A reducing agent is a substance that causes another substance to be reduced.
A reducing agent:
- Removes oxygen from another substance, or
- Loses electrons
It is itself oxidised.
| Term | Oxygen Definition | Electron Definition |
|---|---|---|
| Oxidation | Gain of oxygen | Loss of electrons |
| Reduction | Loss of oxygen | Gain of electrons |
| Oxidising agent | Gives oxygen | Gains electrons |
| Reducing agent | Removes oxygen | Loses electrons |
Example 1 (Conceptual):
Is oxygen oxidised or reduced in the reaction \( \mathrm{2Mg + O_2 \rightarrow 2MgO} \)?
▶️ Answer/Explanation
Oxygen gains electrons.
Therefore oxygen is reduced.
Example 2 (Application):
Identify the reducing agent in \( \mathrm{CuO + Mg \rightarrow Cu + MgO} \).
▶️ Answer/Explanation
Magnesium removes oxygen from copper oxide.
Magnesium is oxidised.
Therefore magnesium is the reducing agent.
Example 3 (Hard):
Explain fully why the reaction between magnesium and copper(II) oxide is a redox reaction using both oxygen and electron definitions.
▶️ Answer/Explanation
Magnesium gains oxygen, so it is oxidised.
Copper(II) oxide loses oxygen, so it is reduced.
Using electron definitions, magnesium loses electrons to form \( \mathrm{Mg^{2+}} \).
Copper ions gain electrons to form copper metal.
Oxidation and reduction occur at the same time.
Therefore the reaction is a redox reaction.
