Edexcel iGCSE Chemistry -2.21 Practical: Metals and Acids- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -2.21 Practical: Metals and Acids- Study Notes- New syllabus
Edexcel iGCSE Chemistry -2.21 Practical: Metals and Acids- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
2.21 practical: investigate reactions between dilute hydrochloric and sulfuric acids and metals (e.g. magnesium, zinc and iron)
2.21 Practical: Reactions Between Dilute Hydrochloric/Sulfuric Acids and Metals
This practical investigates how different metals react with dilute acids and allows comparison of their reactivity.
Metals above hydrogen in the reactivity series react with dilute acids to produce a salt and hydrogen gas.
\( \mathrm{Metal + Acid \rightarrow Salt + Hydrogen} \)
General Equations
With hydrochloric acid: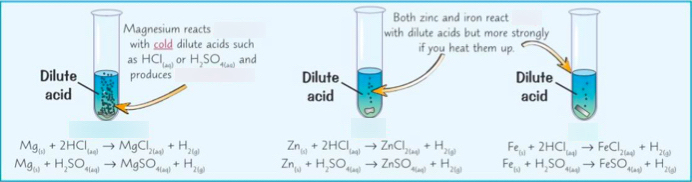
\( \mathrm{Mg(s) + 2HCl(aq) \rightarrow MgCl_2(aq) + H_2(g)} \)
\( \mathrm{Zn(s) + 2HCl(aq) \rightarrow ZnCl_2(aq) + H_2(g)} \)
\( \mathrm{Fe(s) + 2HCl(aq) \rightarrow FeCl_2(aq) + H_2(g)} \)
With sulfuric acid:
\( \mathrm{Mg(s) + H_2SO_4(aq) \rightarrow MgSO_4(aq) + H_2(g)} \)
\( \mathrm{Zn(s) + H_2SO_4(aq) \rightarrow ZnSO_4(aq) + H_2(g)} \)
\( \mathrm{Fe(s) + H_2SO_4(aq) \rightarrow FeSO_4(aq) + H_2(g)} \)
Method
- Measure a fixed volume of dilute acid into a test tube or conical flask.
- Add a known mass or similar-sized piece of metal.
- Observe the reaction (bubbling/fizzing).
- Test gas using a lit splint (squeaky pop confirms hydrogen).
- Compare rate of gas production.
Observations
| Metal | Observation | Relative Reactivity |
|---|---|---|
| Magnesium | Vigorous fizzing | Most reactive |
| Zinc | Moderate fizzing | Medium |
| Iron | Slow bubbling | Least reactive (of these) |
Safety Points
- Wear eye protection.
- Use dilute acids only.
- Keep flames away except when testing gas.
Example 1 (Conceptual):
Why does magnesium react faster than iron with dilute hydrochloric acid?
▶️ Answer/Explanation
Magnesium is higher in the reactivity series.
It loses electrons more easily.
Therefore it reacts more vigorously.
Example 2 (Numerical):
0.50 g of magnesium reacts completely with dilute hydrochloric acid. Calculate the volume of hydrogen gas formed at room temperature and pressure (rtp). (Molar volume at rtp = \( 24\ \mathrm{dm^3} \), Ar(Mg) = 24)
▶️ Answer/Explanation
Moles of Mg:
\( \mathrm{n = \dfrac{0.50}{24} = 0.0208\ mol} \)
From the equation:
\( \mathrm{Mg \rightarrow H_2} \) (1:1 ratio)
Moles of \( \mathrm{H_2} \) = 0.0208 mol
Volume at rtp:
\( \mathrm{Volume = 0.0208 \times 24 = 0.50\ dm^3} \)
Example 3 (Hard):
Describe fully how this practical can be used to arrange magnesium, zinc and iron in order of reactivity.
▶️ Answer/Explanation
Each metal is added separately to dilute acid under controlled conditions.
The rate of hydrogen gas production is observed.
Magnesium reacts most vigorously, producing hydrogen rapidly.
Zinc reacts moderately.
Iron reacts slowly.
The speed of bubbling indicates relative reactivity.
Therefore the order is magnesium, zinc, iron.
This matches their positions in the reactivity series.
