Edexcel iGCSE Chemistry -2.22C Metal Ores and Native Metals- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -2.22C Metal Ores and Native Metals- Study Notes- New syllabus
Edexcel iGCSE Chemistry -2.22C Metal Ores and Native Metals- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
2.22C know that most metals are extracted from ores found in the Earth’s crust and that unreactive metals are often found as the uncombined element
2.22C Extraction of Metals from Ores and Occurrence of Unreactive Metals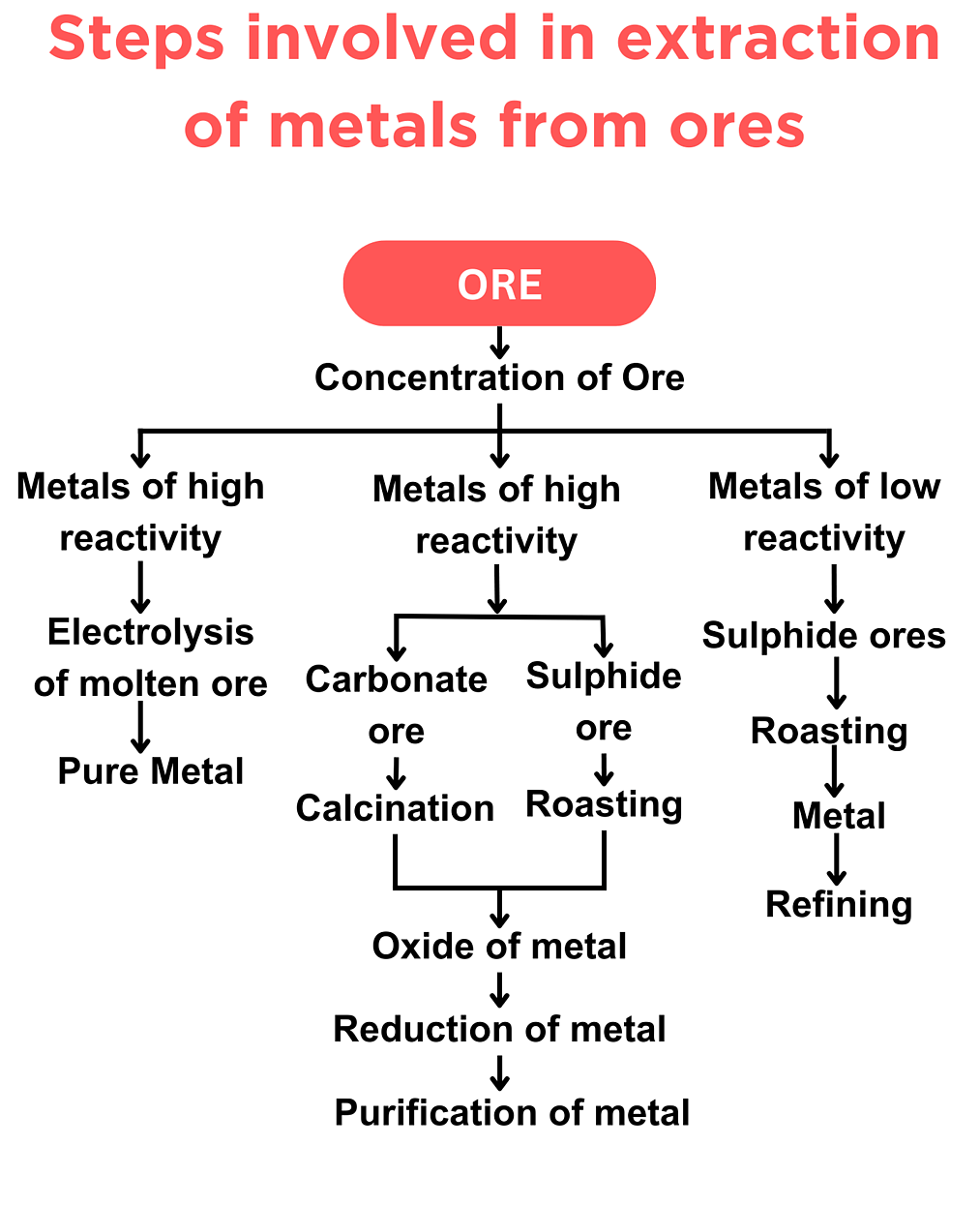
Most metals are too reactive to exist naturally as pure elements.
They are found in the Earth’s crust as compounds called ores.
An ore is a naturally occurring rock that contains enough of a metal compound to make extraction economically worthwhile.
Why Most Metals Are Found as Compounds
- Most metals are reactive.
- They react with oxygen, sulfur or water in the Earth’s crust.
- They form oxides, sulfides or carbonates.
Example: Iron is mainly found as iron oxide in haematite.
\( \mathrm{Fe_2O_3} \)
Example: Aluminium is found as aluminium oxide in bauxite.
\( \mathrm{Al_2O_3} \)
Unreactive Metals
Very unreactive metals do not readily react with oxygen or water.
They are sometimes found as the native (uncombined) element.
Examples:
- Gold (\( \mathrm{Au} \))
- Silver (\( \mathrm{Ag} \))
Gold is very low in the reactivity series, so it does not easily form compounds.
| Metal Type | Reactivity | How Found in Nature | Example |
|---|---|---|---|
| Reactive metals | High | As compounds (ores) | Iron, Aluminium |
| Unreactive metals | Low | As native element | Gold, Silver |
Key Link to Reactivity Series
The position of a metal in the reactivity series determines whether it is found as a compound or as the element.
- High reactivity → found as compounds.
- Low reactivity → may be found uncombined.
Example 1 (Conceptual):
Why is gold often found as the pure element?
▶️ Answer/Explanation
Gold is very unreactive.
It does not easily react with oxygen or water.
Therefore it does not form compounds easily.
Example 2 (Application):
Why is iron found as iron oxide rather than pure iron in the Earth’s crust?
▶️ Answer/Explanation
Iron is reactive.
It reacts with oxygen in the Earth’s crust.
It forms iron oxide compounds.
Example 3 (Hard):
Explain fully why most metals are extracted from ores but gold is often found as the uncombined element.
▶️ Answer/Explanation
Most metals are reactive and lose electrons easily.
They react with oxygen, sulfur or water in the Earth’s crust.
This forms compounds such as oxides or sulfides.
These compounds are found in ores.
Gold is very unreactive and does not readily react with oxygen or water.
Therefore gold remains as the native element in the Earth’s crust.
