Edexcel iGCSE Chemistry -2.23C Metal Extraction Methods- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -2.23C Metal Extraction Methods- Study Notes- New syllabus
Edexcel iGCSE Chemistry -2.23C Metal Extraction Methods- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
2.23C explain how the method of extraction of a metal is related to its position in the reactivity series, illustrated by carbon extraction for iron and electrolysis for aluminium
2.23C Extraction of Metals and the Reactivity Series
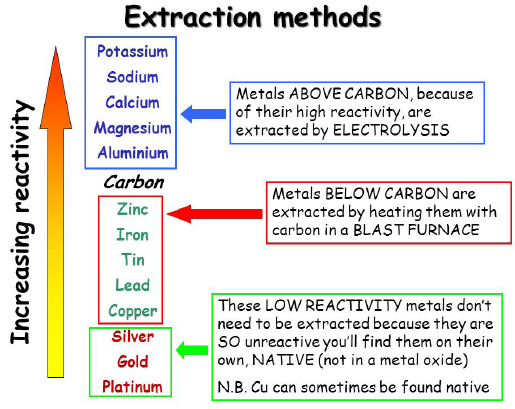
The method used to extract a metal from its ore depends on its position in the reactivity series.
The more reactive a metal is, the more difficult it is to extract.
Key Principle
- Metals below carbon in the reactivity series can be extracted by reduction with carbon.
- Metals above carbon must be extracted using electrolysis.
1. Carbon Extraction – Iron
Iron is below carbon in the reactivity series.
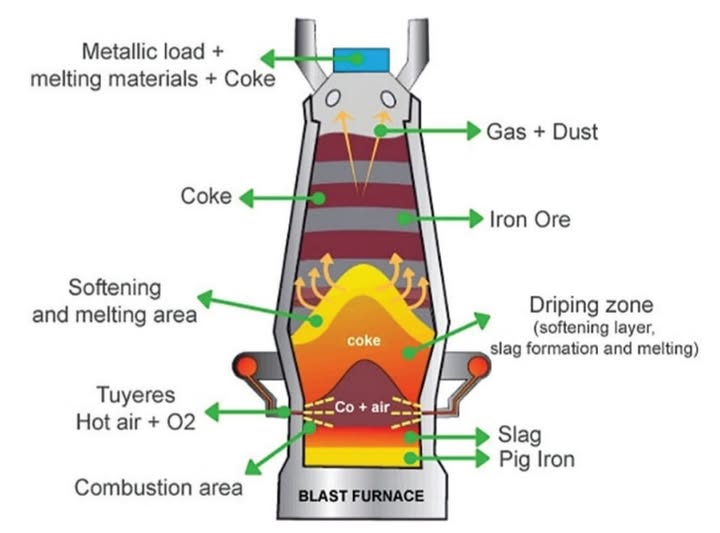
Therefore it can be extracted by reduction using carbon in a blast furnace.
Iron is extracted from iron(III) oxide:
\( \mathrm{Fe_2O_3(s) + 3CO(g) \rightarrow 2Fe(l) + 3CO_2(g)} \)
Carbon monoxide reduces iron(III) oxide to iron.
- Iron oxide loses oxygen → reduced.
- Carbon monoxide gains oxygen → oxidised.
This process is cheaper than electrolysis.
2. Electrolysis – Aluminium
Aluminium is above carbon in the reactivity series.
Carbon cannot remove oxygen from aluminium oxide.
Therefore aluminium is extracted by electrolysis.
Aluminium is extracted from aluminium oxide:
\( \mathrm{Al_2O_3(l) \rightarrow 2Al(l) + \dfrac{3}{2}O_2(g)} \)
At the cathode:
\( \mathrm{Al^{3+} + 3e^- \rightarrow Al} \)
Electrolysis requires large amounts of electricity.
| Metal | Position vs Carbon | Extraction Method | Reason |
|---|---|---|---|
| Iron | Below carbon | Reduction with carbon | Carbon can remove oxygen |
| Aluminium | Above carbon | Electrolysis | Carbon cannot remove oxygen |
Cost Consideration
- Carbon extraction is cheaper.
- Electrolysis is expensive due to electricity costs.
Example 1 (Conceptual):
Why cannot aluminium be extracted using carbon?
▶️ Answer/Explanation
Aluminium is more reactive than carbon.
Carbon cannot remove oxygen from aluminium oxide.
Electrolysis is required instead.
Example 2 (Application):
Why is iron extraction cheaper than aluminium extraction?
▶️ Answer/Explanation
Iron is extracted by carbon reduction.
This uses coke, which is relatively cheap.
Aluminium extraction requires electrolysis, which uses large amounts of electricity.
Example 3 (Hard):
Explain fully how the position of a metal in the reactivity series determines whether it is extracted by carbon reduction or electrolysis.
▶️ Answer/Explanation
Metals below carbon in the reactivity series are less reactive than carbon.
Carbon can remove oxygen from their oxides.
They can therefore be extracted by reduction with carbon.
Metals above carbon are more reactive.
Carbon cannot remove oxygen from their oxides.
They must be extracted by electrolysis.
Electrolysis uses electricity to force the metal ions to gain electrons and form metal.
The position in the reactivity series therefore determines the extraction method.
