Edexcel iGCSE Chemistry -2.24C Metal Extraction Processes- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -2.24C Metal Extraction Processes- Study Notes- New syllabus
Edexcel iGCSE Chemistry -2.24C Metal Extraction Processes- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
2.24C be able to comment on a metal extraction process, given appropriate information
(detailed knowledge of the processes used in the extraction of a specific metal is not required)
2.24C Commenting on a Metal Extraction Process (Using Given Information)
You may be given information about how a metal is extracted and asked to comment on the process.
Detailed knowledge of a specific industrial process is not required.
Instead, you must use:
- The reactivity series
- Energy requirements
- Cost considerations
- Environmental impact
Key Points to Consider
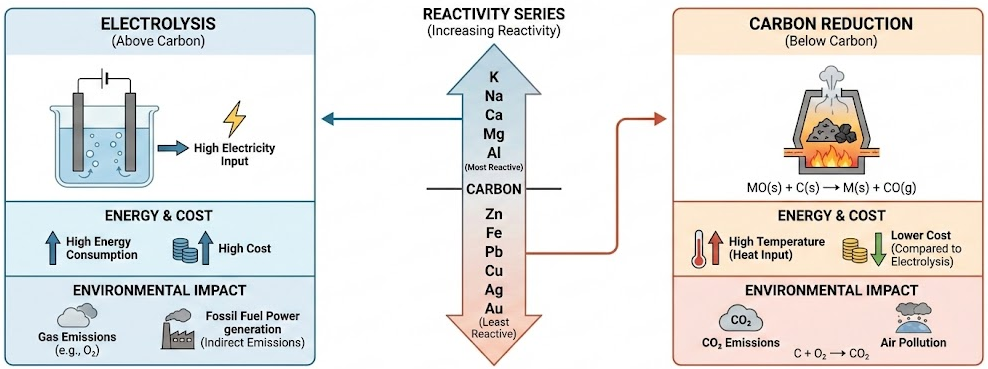
1. Position in the Reactivity Series
- If the metal is below carbon → extraction by carbon reduction is possible.
- If the metal is above carbon → electrolysis is required.
2. Energy Requirements
- Electrolysis requires large amounts of electricity.
- Carbon reduction requires high temperatures but less electrical energy.
Example reduction:
\( \mathrm{MO(s) + C(s) \rightarrow M(s) + CO(g)} \)
3. Environmental Impact
- Carbon reduction produces carbon dioxide:
\( \mathrm{C + O_2 \rightarrow CO_2} \)
- Electrolysis may produce oxygen or other gases.
- Electricity generation may involve fossil fuels.
4. Cost
- Electrolysis is usually more expensive.
- Metals that are very reactive are costly to extract.
- Recycling may be more economical.
| Factor | Carbon Reduction | Electrolysis |
|---|---|---|
| Reactivity | Below carbon | Above carbon |
| Energy cost | High heat | Very high electricity |
| Environmental impact | Produces \( \mathrm{CO_2} \) | Energy source dependent |
Example 1 (Conceptual):
A metal above carbon in the reactivity series is extracted by electrolysis. Explain why.
▶️ Answer/Explanation
The metal is more reactive than carbon.
Carbon cannot remove oxygen from its oxide.
Electrical energy is required instead.
Example 2 (Application):
A process releases large amounts of carbon dioxide during extraction. What method is likely being used?
▶️ Answer/Explanation
Carbon reduction.
Carbon reacts with oxygen to produce \( \mathrm{CO_2} \).
Example 3 (Hard):
A newly discovered metal is more reactive than carbon. Describe and justify how it should be extracted and comment on the likely costs and environmental impact.
▶️ Answer/Explanation
Since the metal is more reactive than carbon, carbon reduction will not work.
Electrolysis must be used to extract the metal from its compound.
Electrolysis requires large amounts of electricity.
This makes the process expensive.
If electricity is generated from fossil fuels, carbon dioxide may be produced.
Therefore the process may have environmental impacts and high energy costs.
