Edexcel iGCSE Chemistry -2.25C Uses of Aluminium, Copper, Iron and Steel- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -Link- Study Notes- New syllabus
Edexcel iGCSE Chemistry -Link- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
2.25C explain the uses of aluminium, copper, iron and steel in terms of their properties
(the types of steel will be limited to low-carbon (mild), high-carbon and stainless)
2.25C Uses of Aluminium, Copper, Iron and Steel in Terms of Their Properties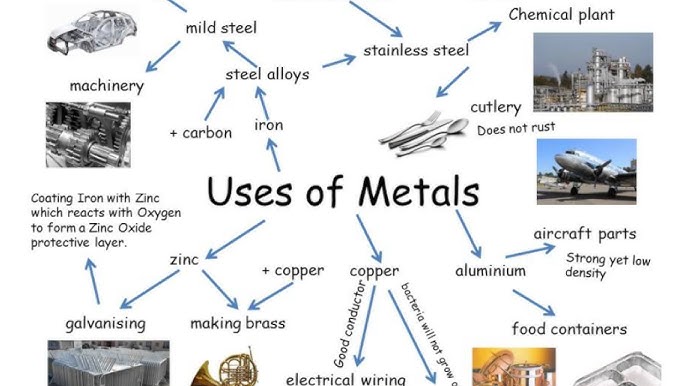
The uses of metals depend on their physical and chemical properties.
Important properties include:
- Density
- Strength
- Hardness
- Electrical conductivity
- Thermal conductivity
- Resistance to corrosion
- Malleability
1. Aluminium
Key properties:
- Low density
- Good conductor of heat and electricity
- Resistant to corrosion (forms protective oxide layer)
- Malleable
Corrosion resistance occurs because aluminium reacts with oxygen to form a thin protective layer:
\( \mathrm{4Al(s) + 3O_2(g) \rightarrow 2Al_2O_3(s)} \)
Uses:
- Aircraft bodies (low density)
- Food containers (corrosion resistant)
- Overhead power cables (low density + conductivity)
2. Copper
Key properties:
- Excellent electrical conductor
- Excellent thermal conductor
- Malleable and ductile
- Resistant to corrosion
Uses:
- Electrical wiring (high conductivity)
- Water pipes (corrosion resistant)
- Cooking utensils (good heat conductor)
3. Iron
Key properties:
- Strong
- Magnetic
- High density
- Prone to rusting
Uses:
- Structural materials (when protected)
- Engine parts
- Tools
4. Steel (Alloy of Iron)
Steel is stronger and harder than pure iron.
Three types are required:
(a) Low-Carbon (Mild) Steel
- Low carbon content
- Malleable
- Relatively cheap
Uses:
- Car bodies
- Construction beams
(b) High-Carbon Steel
- Higher carbon content
- Harder
- Less malleable
- More brittle
Uses:
- Cutting tools
- Drill bits
- Knives
(c) Stainless Steel
- Contains chromium (and often nickel)
- Highly resistant to corrosion
- Strong
Uses:
- Cutlery
- Surgical instruments
- Kitchen equipment
| Material | Key Property | Main Use |
|---|---|---|
| Aluminium | Low density, corrosion resistant | Aircraft |
| Copper | High electrical conductivity | Electrical wiring |
| Low-carbon steel | Malleable | Car bodies |
| High-carbon steel | Hard | Cutting tools |
| Stainless steel | Corrosion resistant | Cutlery |
Example 1 (Conceptual):
Why is aluminium used for aircraft bodies?
▶️ Answer/Explanation
It has low density, so it is lightweight.
It is strong for its mass.
It resists corrosion due to its oxide layer.
Example 2 (Application):
Why is high-carbon steel used for drill bits instead of mild steel?
▶️ Answer/Explanation
High-carbon steel is harder.
It can withstand wear.
Mild steel is softer and would deform.
Example 3 (Hard):
Explain fully why stainless steel is used for cutlery while copper is not.
▶️ Answer/Explanation
Stainless steel is strong and hard.
It contains chromium, which makes it corrosion resistant.
It does not react easily with food or water.
Copper is softer and would scratch easily.
Copper may also react slowly with acidic foods.
Therefore stainless steel is more suitable for cutlery.
