Edexcel iGCSE Chemistry -2.26–2.27C Alloys and Their Properties- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -2.26–2.27C Alloys and Their Properties- Study Notes- New syllabus
Edexcel iGCSE Chemistry -2.26–2.27C Alloys and Their Properties- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
2.26C know that an alloy is a mixture of a metal and one or more elements, usually other metals or carbon
2.27C explain why alloys are harder than pure metals
2.26C Alloys
An alloy is a mixture of:

- A metal
- One or more other elements (usually metals or carbon)
Alloys are made to improve the properties of a metal.
Why Alloys Are Harder Than Pure Metals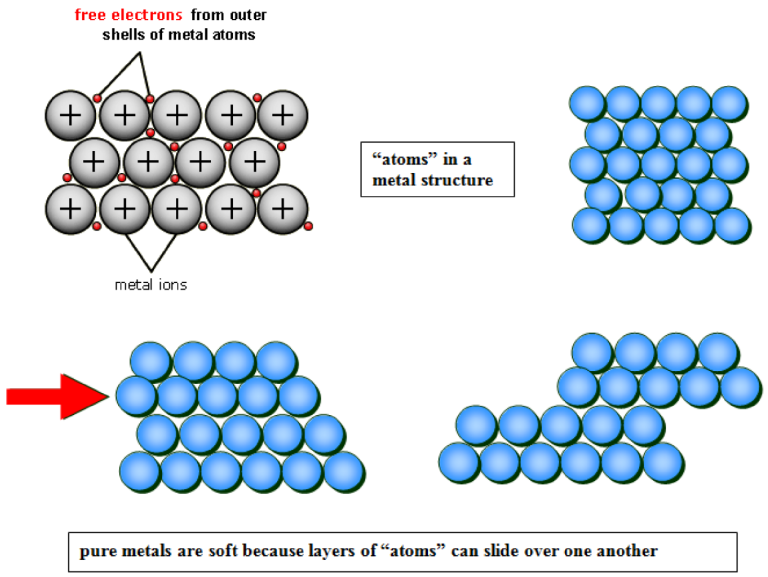
In a pure metal:
- Atoms are arranged in regular layers.
- The layers can slide over each other easily.
- The metal is relatively soft.
In an alloy:
- Atoms of different sizes are mixed together.
- This distorts the regular layers.
- The layers cannot slide easily.
- The alloy is harder and stronger.
Examples of Alloys
Steel
- Iron + carbon
- Stronger than pure iron
Stainless Steel
- Iron + chromium (and often nickel)
- Resistant to corrosion
Brass
- Copper + zinc
- Harder than copper
| Alloy | Composition | Improved Property |
|---|---|---|
| Steel | Iron + carbon | Strength and hardness |
| Stainless steel | Iron + chromium | Corrosion resistance |
| Brass | Copper + zinc | Harder than copper |
Example 1 (Conceptual):
Why is steel harder than pure iron?
▶️ Answer/Explanation
Carbon atoms are different in size to iron atoms.
This distorts the layers of atoms.
The layers cannot slide easily.
The alloy becomes harder.
Example 2 (Application):
Brass is harder than copper. Explain why.
▶️ Answer/Explanation
Brass contains copper and zinc atoms.
The atoms are different sizes.
This prevents layers from sliding easily.
Therefore brass is harder.
Example 3 (Hard ):
Explain fully why alloys are generally harder and stronger than pure metals.
▶️ Answer/Explanation
In pure metals, atoms are arranged in regular layers.
These layers can slide over each other easily.
This makes the metal relatively soft and malleable.
In alloys, atoms of different sizes are mixed.
This distorts the regular arrangement.
The layers cannot slide easily.
Therefore alloys are harder and stronger.
2.27C Why Alloys Are Harder Than Pure Metals
An alloy is a mixture of a metal with one or more other elements (usually metals or carbon).
Alloys are generally harder and stronger than pure metals.
Structure of a Pure Metal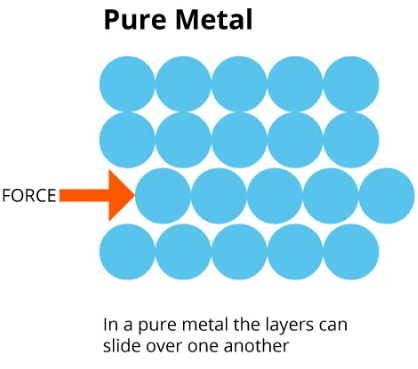
In a pure metal:
- Atoms are arranged in regular layers.
- The atoms are all the same size.
- The layers can slide over each other easily.
Because the layers slide easily, the metal is:
- Soft
- Malleable
Structure of an Alloy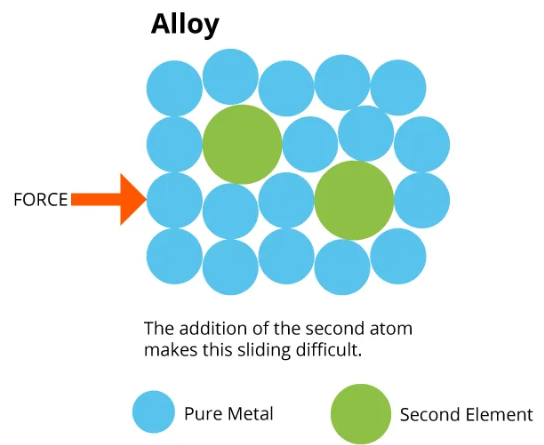
In an alloy:
- Atoms of different elements are mixed.
- The atoms are different sizes.
- This distorts the regular layers.
- The layers cannot slide easily.
As a result:
- The alloy is harder.
- The alloy is stronger.
- It is less malleable.
| Property | Pure Metal | Alloy |
|---|---|---|
| Atom size | All the same | Different sizes |
| Layer movement | Slides easily | Difficult to slide |
| Hardness | Softer | Harder |
Example 1 (Conceptual):
Why is copper softer than brass?
▶️ Answer/Explanation
Copper is a pure metal.
Its layers can slide easily.
Brass contains copper and zinc atoms of different sizes.
This prevents layers from sliding.
Example 2 (Application):
Why is steel stronger than pure iron?
▶️ Answer/Explanation
Steel contains carbon atoms mixed with iron.
The carbon atoms distort the structure.
The layers cannot move easily.
Therefore steel is stronger.
Example 3 (Hard):
Explain fully, in terms of structure, why alloys are harder than pure metals.
▶️ Answer/Explanation
In a pure metal, atoms are arranged in regular layers of identical size.
These layers can slide over each other easily when a force is applied.
This makes the metal soft and malleable.
In an alloy, atoms of different elements and sizes are mixed together.
This disrupts the regular arrangement of atoms.
The layers become distorted.
The layers cannot slide easily.
Therefore alloys are harder and stronger than pure metals.
