Edexcel iGCSE Chemistry -2.28 Indicators- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -2.28 Indicators- Study Notes- New syllabus
Edexcel iGCSE Chemistry -2.28 Indicators- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
2.28 describe the use of litmus, phenolphthalein and methyl orange to distinguish between acidic and alkaline solutions
2.28 Using Indicators to Distinguish Between Acidic and Alkaline Solutions
An indicator is a substance that changes colour depending on the acidity or alkalinity of a solution.
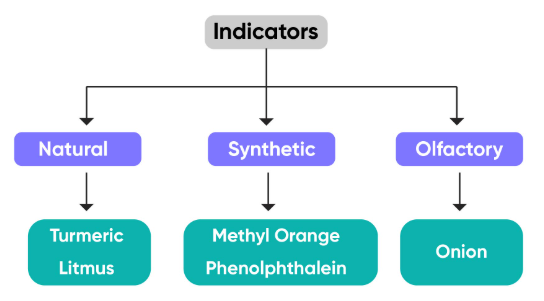
Indicators are used to distinguish between:
- Acidic solutions
- Neutral solutions
- Alkaline solutions
1. Litmus
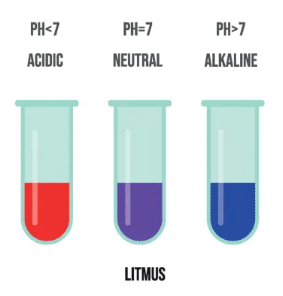
- Red in acids
- Blue in alkalis
- Purple in neutral solutions
Blue litmus turns red in acidic solutions.
Red litmus turns blue in alkaline solutions.
2. Phenolphthalein

- Colourless in acidic solutions
- Colourless in neutral solutions
- Pink in alkaline solutions
Phenolphthalein is useful for detecting alkalis.
3. Methyl Orange
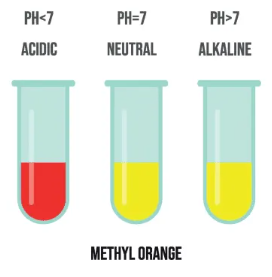
- Red in acidic solutions
- Orange in neutral solutions
- Yellow in alkaline solutions
| Indicator | Acid | Neutral | Alkali |
|---|---|---|---|
| Litmus | Red | Purple | Blue |
| Phenolphthalein | Colourless | Colourless | Pink |
| Methyl orange | Red | Orange | Yellow |
Important Notes
- Indicators do not measure exact pH values.
- They only show whether a solution is acidic, neutral or alkaline.
- Universal indicator is needed to estimate pH more precisely.
Example 1 (Conceptual):
A solution turns phenolphthalein pink. What does this show?
▶️ Answer/Explanation
The solution is alkaline.
Phenolphthalein only turns pink in alkalis.
Example 2 (Application):
A solution turns methyl orange yellow and blue litmus remains blue. What type of solution is it?
▶️ Answer/Explanation
Methyl orange yellow indicates alkaline.
Blue litmus staying blue confirms alkaline.
The solution is alkaline.
Example 3 (Hard):
A colourless solution gives the following results: blue litmus turns red, phenolphthalein remains colourless, methyl orange turns red. Identify the type of solution and explain fully.
▶️ Answer/Explanation
Blue litmus turning red shows the solution is acidic.
Phenolphthalein remaining colourless supports this, since it is colourless in acids.
Methyl orange turning red also indicates an acidic solution.
All three results confirm the solution is acidic.
