Edexcel iGCSE Chemistry -2.29–2.30 The pH Scale, Universal Indicator, and pH- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -2.29–2.30 The pH Scale, Universal Indicator, and pH- Study Notes- New syllabus
Edexcel iGCSE Chemistry -2.29–2.30 The pH Scale, Universal Indicator, and pH- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
2.29 understand how to use the pH scale, from 0–14, to classify solutions as:
• strongly acidic (0–3)
• weakly acidic (4–6)
• neutral (7)
• weakly alkaline (8–10)
• strongly alkaline (11–14)
2.30 describe the use of universal indicator to measure the approximate pH value of an aqueous solution
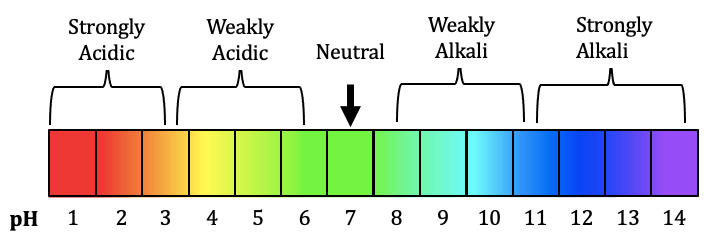
2.29 The pH Scale (0–14)
The pH scale is used to measure how acidic or alkaline a solution is.
The scale runs from 0 to 14.
pH is related to the concentration of hydrogen ions in solution.
\( \mathrm{pH = -\log_{10}[H^+]} \)
Lower pH → higher \( \mathrm{[H^+]} \) → more acidic.
Classification of Solutions Using the pH Scale
| pH Range | Classification | Description |
|---|---|---|
| 0–3 | Strongly acidic | High concentration of \( \mathrm{H^+} \) |
| 4–6 | Weakly acidic | Lower concentration of \( \mathrm{H^+} \) |
| 7 | Neutral | Equal \( \mathrm{[H^+]} \) and \( \mathrm{[OH^-]} \) |
| 8–10 | Weakly alkaline | Lower concentration of \( \mathrm{OH^-} \) |
| 11–14 | Strongly alkaline | High concentration of \( \mathrm{OH^-} \) |
Key Points About the pH Scale
- Each whole number change in pH represents a 10× change in \( \mathrm{[H^+]} \).
- pH 3 is 10 times more acidic than pH 4.
- pH 2 is 100 times more acidic than pH 4.
At pH 7:
\( \mathrm{[H^+] = [OH^-]} \)
Examples of Substances
- Strong acid (e.g. hydrochloric acid) → pH 0–2
- Weak acid (e.g. ethanoic acid) → pH 3–6
- Pure water → pH 7
- Weak alkali (e.g. ammonia solution) → pH 8–10
- Strong alkali (e.g. sodium hydroxide) → pH 11–14
Example 1 (Conceptual):
A solution has a pH of 2. How would you classify it?
▶️ Answer/Explanation
pH 2 falls in the range 0–3.
The solution is strongly acidic.
Example 2 (Application):
Which solution is more acidic: pH 3 or pH 5? Explain.
▶️ Answer/Explanation
pH 3 is more acidic.
Each pH unit represents a 10× change.
pH 3 has 100 times more \( \mathrm{H^+} \) than pH 5.
Example 3 (Hard):
A solution has pH 11. Explain fully what this tells you about its acidity or alkalinity.
▶️ Answer/Explanation
pH 11 is in the range 11–14.
This means the solution is strongly alkaline.
The concentration of \( \mathrm{OH^-} \) ions is high.
The concentration of \( \mathrm{H^+} \) ions is low.
Therefore the solution is a strong alkali.
2.30 Using Universal Indicator to Measure the Approximate pH of an Aqueous Solution
A universal indicator is a mixture of several indicators.
It produces a range of colours across the pH scale from 0 to 14.
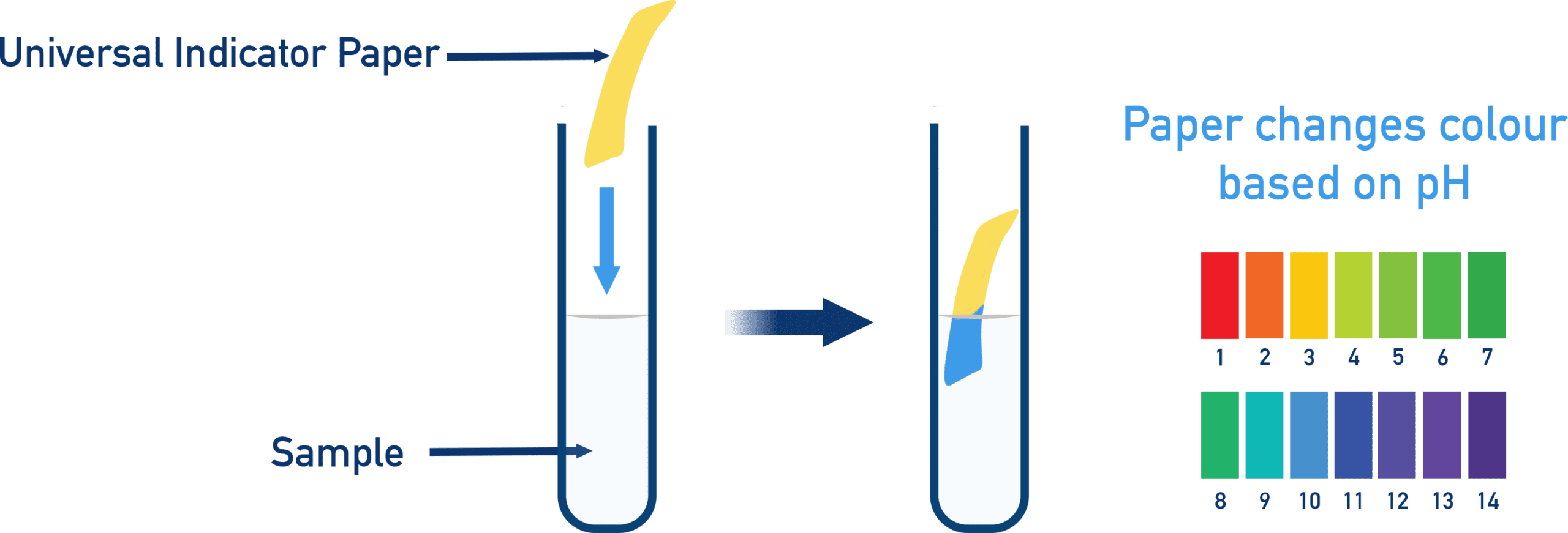
Unlike single indicators (e.g. litmus), universal indicator gives an approximate numerical pH value.
How Universal Indicator Works
The colour produced depends on the concentration of hydrogen ions in solution.
Higher \( \mathrm{[H^+]} \) → lower pH → red/orange colour
Lower \( \mathrm{[H^+]} \) → higher pH → blue/purple colour
Colour Changes of Universal Indicator
| pH | Colour | Type of Solution |
|---|---|---|
| 0–2 | Red | Strong acid |
| 3–4 | Orange | Acid |
| 5–6 | Yellow | Weak acid |
| 7 | Green | Neutral |
| 8–10 | Blue | Alkali |
| 11–14 | Purple | Strong alkali |
Method of Use
- Add a few drops of universal indicator solution to the aqueous solution.
- Observe the colour change.
- Compare the colour with a pH colour chart.
- Estimate the pH value.
Universal indicator can also be used in paper form (pH paper).
Limitations
- It gives an approximate pH only.
- It is not precise to decimal places.
- A pH meter is needed for accurate measurements.
Example 1 (Conceptual):
A solution turns universal indicator green. What is its approximate pH?
▶️ Answer/Explanation
Green corresponds to pH 7.
The solution is neutral.
Example 2 (Application):
A solution turns universal indicator blue. Is it acidic or alkaline? Estimate the pH.
▶️ Answer/Explanation
Blue indicates an alkaline solution.
The approximate pH is between 8 and 10.
Example 3 (Hard):
A colourless solution turns universal indicator orange. Explain fully what this shows about the solution.
▶️ Answer/Explanation
Orange corresponds to a pH around 3–4.
This means the solution is acidic.
The concentration of \( \mathrm{H^+} \) ions is relatively high.
It is likely a weak to moderately strong acid.
The pH value is approximate.
