Edexcel iGCSE Chemistry -2.32 Neutralisation- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -2.32 Neutralisation- Study Notes- New syllabus
Edexcel iGCSE Chemistry -2.32 Neutralisation- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
2.32 know that alkalis can neutralise acids
2.32 Alkalis Neutralise Acids
An alkali is a soluble base that produces hydroxide ions, \( \mathrm{OH^-} \), in aqueous solution.
An acid produces hydrogen ions, \( \mathrm{H^+} \), in aqueous solution.
When an acid reacts with an alkali, they neutralise each other.
Definition of Neutralisation![]()
Neutralisation is the reaction between:
- An acid
- An alkali (or base)
to form:
- A salt
- Water
\( \mathrm{Acid + Alkali \rightarrow Salt + Water} \)
Ionic Equation for Neutralisation
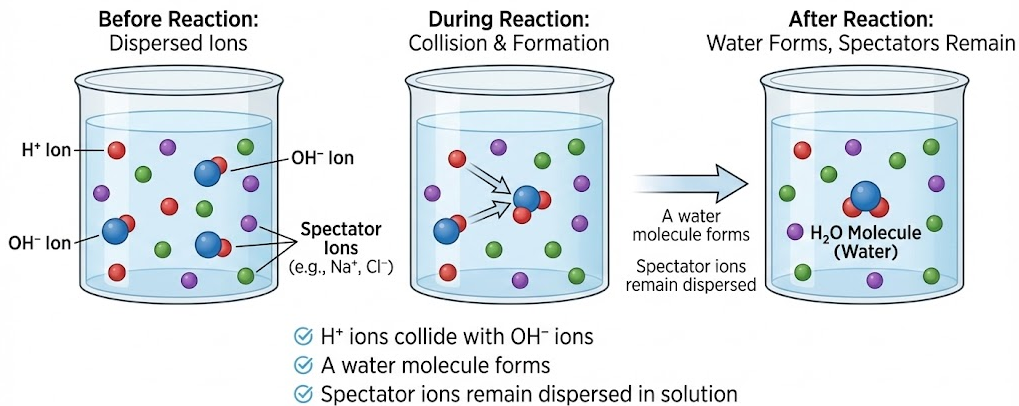
\( \mathrm{H^+(aq) + OH^-(aq) \rightarrow H_2O(l)} \)
Hydrogen ions from the acid react with hydroxide ions from the alkali to form water.
Example Reactions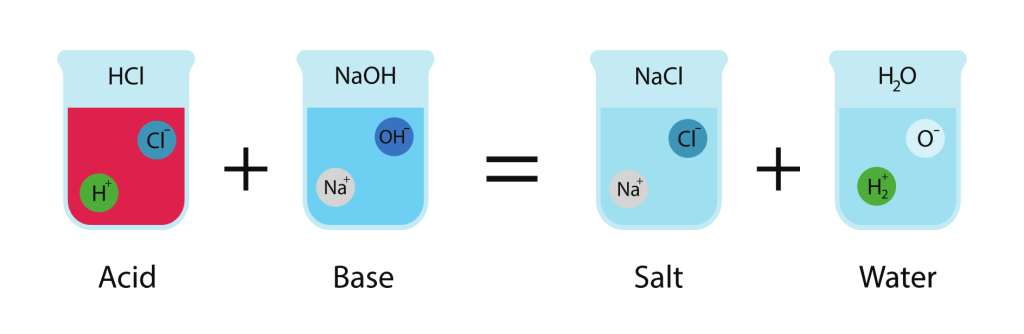
Hydrochloric acid and sodium hydroxide:
\( \mathrm{HCl(aq) + NaOH(aq) \rightarrow NaCl(aq) + H_2O(l)} \)
Sulfuric acid and potassium hydroxide:
\( \mathrm{H_2SO_4(aq) + 2KOH(aq) \rightarrow K_2SO_4(aq) + 2H_2O(l)} \)
Effects on pH
- If equal amounts react, the final solution becomes neutral (pH 7).
- If excess acid remains, the solution stays acidic.
- If excess alkali remains, the solution stays alkaline.
Example 1 (Conceptual):
Why does sodium hydroxide neutralise hydrochloric acid?
▶️ Answer/Explanation
Sodium hydroxide provides \( \mathrm{OH^-} \) ions.
Hydrochloric acid provides \( \mathrm{H^+} \) ions.
\( \mathrm{H^+} \) reacts with \( \mathrm{OH^-} \) to form water.
This removes the acidic properties.
Example 2 (Application):
If a solution changes from pH 2 to pH 7 after adding an alkali, what has happened?
▶️ Answer/Explanation
The alkali neutralised the acid.
\( \mathrm{H^+} \) ions reacted with \( \mathrm{OH^-} \) ions to form water.
The solution became neutral.
Example 3 (Hard):
Explain fully, using ionic equations, how an alkali neutralises an acid.
▶️ Answer/Explanation
Acids produce \( \mathrm{H^+} \) ions in aqueous solution.
Alkalis produce \( \mathrm{OH^-} \) ions in aqueous solution.
These ions react according to:
\( \mathrm{H^+ + OH^- \rightarrow H_2O} \)
Water is formed.
The removal of hydrogen ions means the solution is no longer acidic.
The reaction produces a salt and water.
This process is neutralisation.
