Edexcel iGCSE Chemistry -2.33C Acid–Alkali Titration- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -2.33C Acid–Alkali Titration- Study Notes- New syllabus
Edexcel iGCSE Chemistry -2.33C Acid–Alkali Titration- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
2.33C describe how to carry out an acid–alkali titration
2.33C Acid–Alkali Titration
An acid–alkali titration is used to determine the concentration of an acid or an alkali.
It involves carefully measuring the volume of one solution required to neutralise a known volume of another solution.
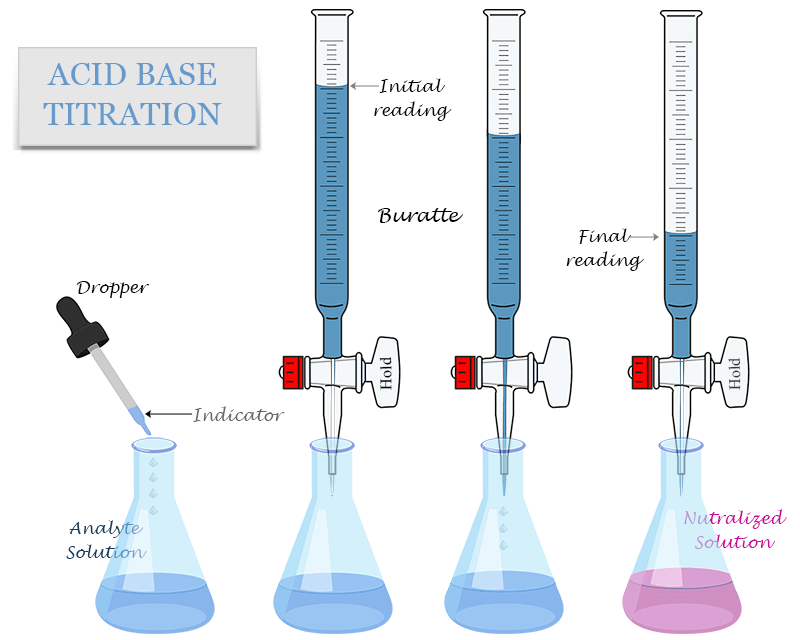
Principle of Titration
\( \mathrm{Acid + Alkali \rightarrow Salt + Water} \)
Neutralisation occurs when:
\( \mathrm{H^+(aq) + OH^-(aq) \rightarrow H_2O(l)} \)
The end-point is when exactly enough acid has been added to react with all the alkali (or vice versa).
Apparatus Used
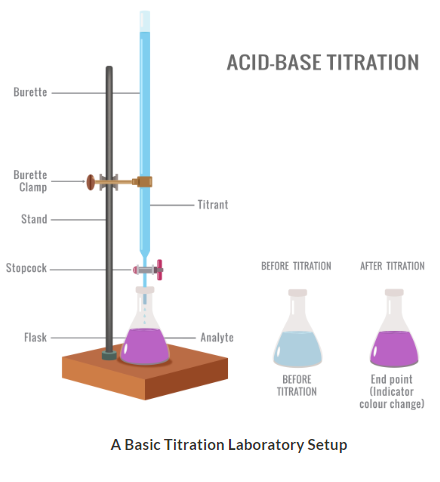
- Burette (for accurate volume measurement)
- Pipette (to measure fixed volume of solution)
- Conical flask
- White tile (to see colour change clearly)
- Indicator (e.g. phenolphthalein or methyl orange)
Method
- Rinse the burette with the solution to be placed inside it.
- Fill the burette with the acid (or alkali) and record the initial volume.
- Use a pipette to transfer a measured volume of the other solution into a conical flask.
- Add a few drops of suitable indicator.
- Place the flask under the burette.
- Slowly add solution from the burette while swirling the flask.
- Stop adding when the indicator just changes colour permanently.
- Record the final burette reading.
- Calculate the volume added (final reading − initial reading).
- Repeat to obtain concordant results.
Choice of Indicator
- Phenolphthalein: colourless in acid, pink in alkali.
- Methyl orange: red in acid, yellow in alkali.
The correct indicator depends on the acid–alkali combination.
Accuracy Tips
- Read the bottom of the meniscus at eye level.
- Add solution drop by drop near the end-point.
- Repeat until two concordant results (within 0.1 cm³).
Example 1 (Conceptual):
Why is the conical flask placed on a white tile?
▶️ Answer/Explanation
To see the colour change of the indicator clearly.
The white background makes the end-point easier to observe.
Example 2 (Application):
The initial burette reading is 2.30 cm³ and the final reading is 27.80 cm³. Calculate the volume added.
▶️ Answer/Explanation
\( \mathrm{Volume = 27.80 – 2.30 = 25.50\ cm^3} \)
Example 3 (Hard):
Describe fully how you would carry out an acid–alkali titration to obtain accurate results.
▶️ Answer/Explanation
Rinse the burette with the solution it will contain.
Fill the burette and record the initial reading at eye level.
Use a pipette to measure a fixed volume of the other solution into a conical flask.
Add a few drops of indicator.
Place the flask under the burette on a white tile.
Add solution slowly while swirling continuously.
Near the end-point, add drop by drop.
Stop when the indicator just changes colour permanently.
Record the final reading.
Repeat the titration until concordant results are obtained.
