Edexcel iGCSE Chemistry -2.34 Solubility Rules- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -2.34 Solubility Rules- Study Notes- New syllabus
Edexcel iGCSE Chemistry -2.34 Solubility Rules- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
2.34 know the general rules for predicting the solubility of ionic compounds in water
2.34 General Rules for Predicting the Solubility of Ionic Compounds in Water
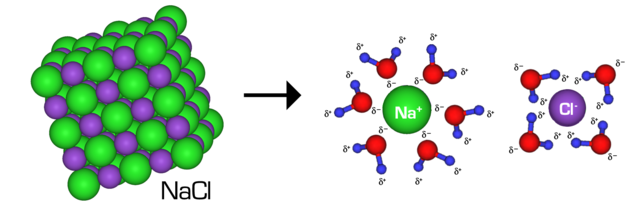
Solubility rules allow us to predict whether an ionic compound will dissolve in water.
If a compound is soluble, it forms an aqueous solution:
\( \mathrm{Compound(s) \rightarrow Ions(aq)} \)
If a compound is insoluble, it forms a solid precipitate.
Important Solubility Rules
| Type of Compound | Solubility Rule |
|---|---|
| Sodium, potassium, ammonium compounds | All soluble |
| Nitrates | All soluble |
| Chlorides | Soluble except \( \mathrm{AgCl} \) and \( \mathrm{PbCl_2} \) |
| Sulfates | Soluble except \( \mathrm{BaSO_4} \), \( \mathrm{CaSO_4} \), \( \mathrm{PbSO_4} \) |
| Carbonates | Insoluble except Na, K, NH₄ compounds |
| Hydroxides | Insoluble except NaOH, KOH and Ca(OH)₂ (slightly soluble) |
Understanding Exceptions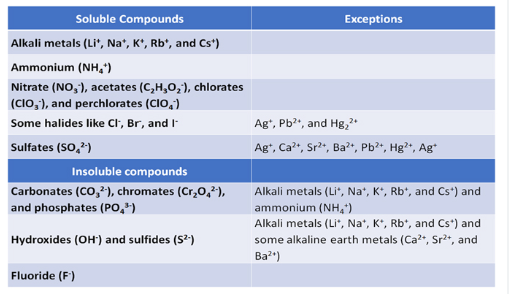
- Silver chloride is insoluble:
\( \mathrm{AgCl(s)} \)
- Barium sulfate is insoluble:
\( \mathrm{BaSO_4(s)} \)
- Calcium hydroxide is only slightly soluble:
\( \mathrm{Ca(OH)_2(aq)} \) (limited solubility)
Key Idea
If a compound contains:
- \( \mathrm{Na^+} \), \( \mathrm{K^+} \), or \( \mathrm{NH_4^+} \) → it is soluble.
- \( \mathrm{NO_3^-} \) → it is soluble.
These rules are heavily tested in precipitation reactions.
Example 1 (Conceptual):
Is potassium carbonate soluble in water? Explain.
▶️ Answer/Explanation
Yes.
All potassium compounds are soluble.
Therefore potassium carbonate dissolves in water.
Example 2 (Application):
Predict whether silver nitrate and sodium chloride will form a precipitate when mixed.
▶️ Answer/Explanation
Silver nitrate is soluble.
Sodium chloride is soluble.
Silver chloride is an exception and is insoluble:
\( \mathrm{Ag^+(aq) + Cl^-(aq) \rightarrow AgCl(s)} \)
A white precipitate of \( \mathrm{AgCl} \) forms.
Example 3 (Hard):
Predict whether a precipitate forms when aqueous barium chloride is mixed with aqueous sodium sulfate. Explain fully.
▶️ Answer/Explanation
Barium chloride is soluble.
Sodium sulfate is soluble.
When mixed, the possible products are barium sulfate and sodium chloride.
Sodium chloride is soluble.
Barium sulfate is an exception and is insoluble.
The ionic equation is:
\( \mathrm{Ba^{2+}(aq) + SO_4^{2-}(aq) \rightarrow BaSO_4(s)} \)
A white precipitate of \( \mathrm{BaSO_4} \) forms.
