Edexcel iGCSE Chemistry -2.35–2.36 Acids and Bases as Proton Transfer (Brønsted–Lowry Theory)- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -2.35–2.36 Acids and Bases as Proton Transfer (Brønsted–Lowry Theory)- Study Notes- New syllabus
Edexcel iGCSE Chemistry -2.35–2.36 Acids and Bases as Proton Transfer (Brønsted–Lowry Theory)- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
2.35 understand acids and bases in terms of proton transfer
2.36 understand that an acid is a proton donor and a base is a proton acceptor
2.35 Acids and Bases in Terms of Proton Transfer
The proton transfer definition of acids and bases is known as the Brønsted–Lowry theory.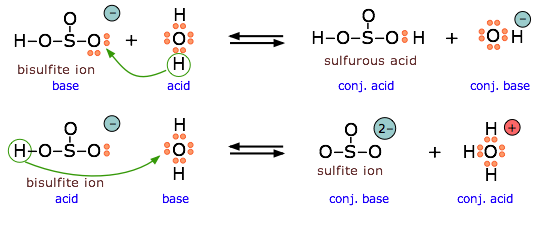
In this theory:
- An acid is a proton donor.
- A base is a proton acceptor.
A proton is a hydrogen ion:
\( \mathrm{H^+} \)
Acid–Base Reaction as Proton Transfer
Example: Reaction between hydrochloric acid and ammonia.
\( \mathrm{HCl(aq) + NH_3(aq) \rightarrow NH_4^+(aq) + Cl^-(aq)} \)
- \( \mathrm{HCl} \) donates a proton → acid.
- \( \mathrm{NH_3} \) accepts a proton → base.
The proton \( \mathrm{H^+} \) is transferred from the acid to the base.
Example in Water
When hydrochloric acid dissolves in water:
\( \mathrm{HCl(aq) + H_2O(l) \rightarrow H_3O^+(aq) + Cl^-(aq)} \)
- \( \mathrm{HCl} \) donates a proton.
- \( \mathrm{H_2O} \) accepts a proton.
Water acts as a base in this reaction.
Neutralisation as Proton Transfer
In a neutralisation reaction:
\( \mathrm{H^+(aq) + OH^-(aq) \rightarrow H_2O(l)} \)
- The acid provides the proton.
- The hydroxide ion accepts the proton.
This is a proton transfer reaction.
| Term | Definition (Proton Transfer) |
|---|---|
| Acid | Proton donor |
| Base | Proton acceptor |
Example 1 (Conceptual):
Why is ammonia a base in the reaction with hydrochloric acid?
▶️ Answer/Explanation
Ammonia accepts a proton \( \mathrm{H^+} \).
It forms \( \mathrm{NH_4^+} \).
Therefore it is a proton acceptor.
Example 2 (Application):
In the reaction \( \mathrm{HCl + H_2O \rightarrow H_3O^+ + Cl^-} \), identify the acid and the base.
▶️ Answer/Explanation
\( \mathrm{HCl} \) donates a proton → acid.
\( \mathrm{H_2O} \) accepts a proton → base.
Example 3 (Hard):
Explain fully why neutralisation is described as a proton transfer reaction.
▶️ Answer/Explanation
An acid produces \( \mathrm{H^+} \) ions in aqueous solution.
An alkali produces \( \mathrm{OH^-} \) ions in aqueous solution.
The hydrogen ion is transferred from the acid to the hydroxide ion.
This forms water:
\( \mathrm{H^+ + OH^- \rightarrow H_2O} \)
The transfer of a proton from acid to base defines the reaction as proton transfer.
2.36 Acids and Bases as Proton Donors and Proton Acceptors
According to the Brønsted–Lowry theory:
- An acid is a proton donor.
- A base is a proton acceptor.
A proton is a hydrogen ion:
\( \mathrm{H^+} \)
How a Proton Is Donated
When an acid donates a proton, it loses \( \mathrm{H^+} \).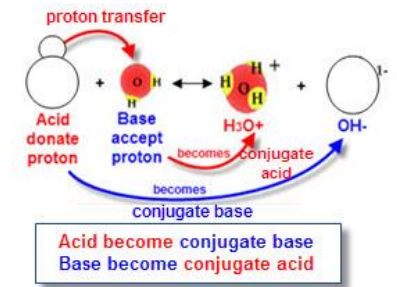
Example:
\( \mathrm{HCl(aq) \rightarrow H^+(aq) + Cl^-(aq)} \)
- \( \mathrm{HCl} \) gives away a proton.
- Therefore, \( \mathrm{HCl} \) is an acid.
How a Proton Is Accepted
When a base accepts a proton, it gains \( \mathrm{H^+} \).
Example:
\( \mathrm{NH_3(aq) + H^+(aq) \rightarrow NH_4^+(aq)} \)
- \( \mathrm{NH_3} \) accepts a proton.
- Therefore, \( \mathrm{NH_3} \) is a base.
Example Reaction Showing Both
\( \mathrm{HCl(aq) + NH_3(aq) \rightarrow NH_4^+(aq) + Cl^-(aq)} \)
- \( \mathrm{HCl} \) donates a proton → acid.
- \( \mathrm{NH_3} \) accepts a proton → base.
The reaction is a proton transfer reaction.
| Substance | Role | Reason |
|---|---|---|
| \( \mathrm{HCl} \) | Acid | Donates \( \mathrm{H^+} \) |
| \( \mathrm{NH_3} \) | Base | Accepts \( \mathrm{H^+} \) |
Example 1 (Conceptual):
Why is hydrochloric acid described as a proton donor?
▶️ Answer/Explanation
It releases \( \mathrm{H^+} \) ions in aqueous solution.
Therefore, it donates protons.
Example 2 (Application):
Identify the acid and base in \( \mathrm{NH_3 + H^+ \rightarrow NH_4^+} \).
▶️ Answer/Explanation
\( \mathrm{NH_3} \) is the base because it accepts a proton.
\( \mathrm{H^+} \) comes from the acid.
Example 3 (Hard ):
Explain fully why the reaction between hydrochloric acid and ammonia is an acid–base reaction in terms of proton transfer.
▶️ Answer/Explanation
Hydrochloric acid donates a proton \( \mathrm{H^+} \).
Ammonia accepts this proton.
This forms ammonium ions \( \mathrm{NH_4^+} \).
Because one substance donates a proton and the other accepts it, the reaction fits the definition of an acid–base reaction according to the Brønsted–Lowry theory.
