Edexcel iGCSE Chemistry -2.37 Reactions of Acids- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -2.37 Reactions of Acids- Study Notes- New syllabus
Edexcel iGCSE Chemistry -2.37 Reactions of Acids- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
2.37 describe the reactions of hydrochloric acid, sulfuric acid and nitric acid with metals, bases and metal carbonates (excluding nitric acid with metals) to form salts
2.37 Reactions of Hydrochloric, Sulfuric and Nitric Acids to Form Salts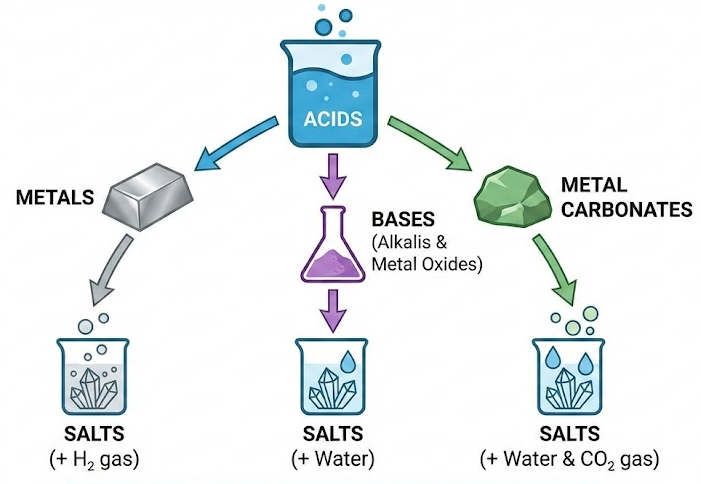
Acids react with:
- Metals
- Bases (including alkalis and metal oxides)
- Metal carbonates
to form salts.
The type of salt formed depends on the acid used.
Salts Formed by Different Acids
| Acid | Salt Formed |
|---|---|
| Hydrochloric acid \( \mathrm{HCl} \) | Chlorides |
| Sulfuric acid \( \mathrm{H_2SO_4} \) | Sulfates |
| Nitric acid \( \mathrm{HNO_3} \) | Nitrates |
1. Reactions with Metals (Except Nitric Acid)
General equation:
\( \mathrm{Metal + Acid \rightarrow Salt + Hydrogen} \)
Example with hydrochloric acid:
\( \mathrm{Mg + 2HCl \rightarrow MgCl_2 + H_2} \)
Example with sulfuric acid:
\( \mathrm{Zn + H_2SO_4 \rightarrow ZnSO_4 + H_2} \)
Note: Nitric acid reacting with metals is excluded.
2. Reactions with Bases
Bases include:
- Metal oxides
- Metal hydroxides (alkalis)
General equation:
\( \mathrm{Acid + Base \rightarrow Salt + Water} \)
Example:
\( \mathrm{CuO + 2HCl \rightarrow CuCl_2 + H_2O} \)
\( \mathrm{NaOH + HCl \rightarrow NaCl + H_2O} \)
3. Reactions with Metal Carbonates
General equation:
\( \mathrm{Acid + Metal\ Carbonate \rightarrow Salt + Water + Carbon\ Dioxide} \)
Example:
\( \mathrm{CaCO_3 + 2HCl \rightarrow CaCl_2 + H_2O + CO_2} \)
\( \mathrm{Na_2CO_3 + H_2SO_4 \rightarrow Na_2SO_4 + H_2O + CO_2} \)
Summary of Products
| Reactants | Products |
|---|---|
| Acid + Metal | Salt + Hydrogen |
| Acid + Base | Salt + Water |
| Acid + Metal carbonate | Salt + Water + Carbon dioxide |
Example 1 (Conceptual):
What type of salt is formed when sulfuric acid reacts?
▶️ Answer/Explanation
A sulfate is formed.
Sulfuric acid produces sulfate salts.
Example 2 (Application):
Write the balanced equation for magnesium reacting with hydrochloric acid.
▶️ Answer/Explanation
\( \mathrm{Mg + 2HCl \rightarrow MgCl_2 + H_2} \)
Example 3 (Hard):
Describe fully the products formed when calcium carbonate reacts with nitric acid.
▶️ Answer/Explanation
Nitric acid forms nitrate salts.
The general reaction of an acid with a metal carbonate produces a salt, water and carbon dioxide.
The balanced equation is:
\( \mathrm{CaCO_3 + 2HNO_3 \rightarrow Ca(NO_3)_2 + H_2O + CO_2} \)
The products are calcium nitrate, water and carbon dioxide.
