Edexcel iGCSE Chemistry -2.38 Bases and Alkalis- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -2.38 Bases and Alkalis- Study Notes- New syllabus
Edexcel iGCSE Chemistry -2.38 Bases and Alkalis- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
2.38 know that metal oxides, metal hydroxides and ammonia can act as bases, and that alkalis are bases that are soluble in water
2.38 Bases and Alkalis
A base is a substance that neutralises an acid.
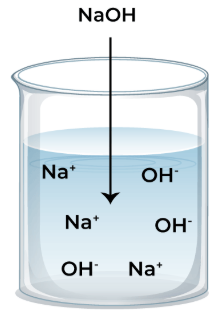
An alkali is a base that is soluble in water and produces hydroxide ions, \( \mathrm{OH^-} \), in aqueous solution.
1. Metal Oxides as Bases
Most metal oxides are basic.
They react with acids to form a salt and water.
\( \mathrm{Metal\ oxide + Acid \rightarrow Salt + Water} \)
Example:
\( \mathrm{CuO + 2HCl \rightarrow CuCl_2 + H_2O} \)
Copper(II) oxide acts as a base because it neutralises hydrochloric acid.
2. Metal Hydroxides as Bases
Metal hydroxides contain the hydroxide ion \( \mathrm{OH^-} \).
They react with acids to form salt and water.
\( \mathrm{NaOH + HCl \rightarrow NaCl + H_2O} \)
Some metal hydroxides are soluble (alkalis), others are insoluble.
3. Ammonia as a Base
Ammonia, \( \mathrm{NH_3} \), is a base.
In water, it reacts to produce hydroxide ions:
\( \mathrm{NH_3(aq) + H_2O(l) \rightleftharpoons NH_4^+(aq) + OH^-(aq)} \)
Because it produces \( \mathrm{OH^-} \) ions in water, ammonia solution is an alkali.
Difference Between Bases and Alkalis
| Term | Definition | Example |
|---|---|---|
| Base | Neutralises an acid | CuO, MgO |
| Alkali | Soluble base producing \( \mathrm{OH^-} \) | NaOH, KOH, NH₃(aq) |
Key Idea
- All alkalis are bases.
- Not all bases are alkalis.
For example, copper(II) oxide is a base but is not soluble, so it is not an alkali.
Example 1 (Conceptual):
Why is sodium hydroxide both a base and an alkali?
▶️ Answer/Explanation
It neutralises acids, so it is a base.
It is soluble in water and produces \( \mathrm{OH^-} \) ions.
Therefore it is also an alkali.
Example 2 (Application):
Is magnesium oxide an alkali? Explain.
▶️ Answer/Explanation
Magnesium oxide is a base because it neutralises acids.
However, it is not soluble in water.
Therefore it is not an alkali.
Example 3 (Hard):
Explain fully why ammonia solution is an alkali even though it does not contain the hydroxide ion in its formula.
▶️ Answer/Explanation
Ammonia does not contain \( \mathrm{OH^-} \) in its formula.
However, when dissolved in water, it reacts with water molecules.
The reaction forms ammonium ions and hydroxide ions:
\( \mathrm{NH_3 + H_2O \rightleftharpoons NH_4^+ + OH^-} \)
Because hydroxide ions are produced in solution, ammonia solution is alkaline.
Since it is soluble, it is classified as an alkali.
