Edexcel iGCSE Chemistry -2.39–2.41C Preparation of Soluble and Insoluble Salts- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -2.39–2.41C Preparation of Soluble and Insoluble Salts- Study Notes- New syllabus
Edexcel iGCSE Chemistry -2.39–2.41C Preparation of Soluble and Insoluble Salts- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
2.39 describe an experiment to prepare a pure, dry sample of a soluble salt, starting from an insoluble reactant
2.40C describe an experiment to prepare a pure, dry sample of a soluble salt, starting from an acid and alkali
2.41C describe an experiment to prepare a pure, dry sample of an insoluble salt, starting from two soluble reactants
2.39 Preparing a Pure, Dry Sample of a Soluble Salt (From an Insoluble Reactant)
A soluble salt can be prepared by reacting an insoluble base (such as a metal oxide or metal carbonate) with an acid.
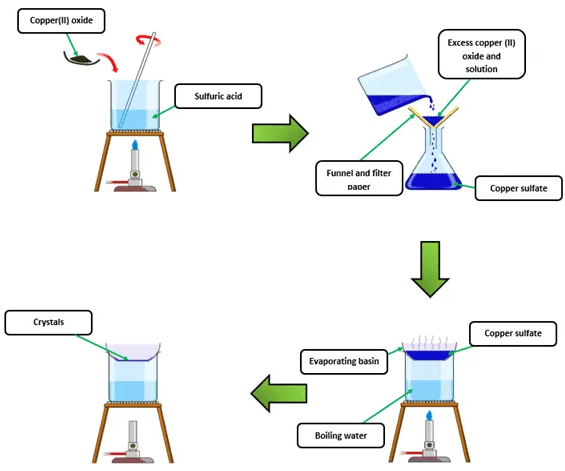
Excess insoluble solid is used to ensure all the acid is neutralised.
Example Reaction
Preparing copper(II) sulfate from copper(II) oxide and sulfuric acid:
\( \mathrm{CuO(s) + H_2SO_4(aq) \rightarrow CuSO_4(aq) + H_2O(l)} \)
Copper(II) oxide is insoluble, so it can be filtered off easily.
Method
- Measure a fixed volume of dilute acid into a beaker.
- Gently heat the acid using a Bunsen burner (do not boil).
- Add the insoluble solid (e.g. copper(II) oxide) in small portions while stirring.
- Continue adding until some solid remains unreacted (excess).
- Filter the mixture to remove excess solid.
- Collect the filtrate (salt solution) in an evaporating basin.
- Gently heat to evaporate some water.
- Allow the solution to cool so crystals form.
- Filter the crystals and dry them between filter papers.
Why Excess Solid Is Used
- Ensures all acid is completely neutralised.
- Any remaining solid can be removed by filtration.
- This prevents acid contamination of the salt.
Key Stages
| Stage | Purpose |
|---|---|
| Add excess insoluble solid | Neutralise all acid |
| Filtration | Remove excess solid |
| Evaporation | Concentrate solution |
| Crystallisation | Form pure crystals |
Example 1 (Conceptual):
Why is filtration necessary in this method?
▶️ Answer/Explanation
Filtration removes the excess insoluble solid.
This leaves a pure salt solution.
Example 2 (Application):
What would happen if insufficient copper(II) oxide were added?
▶️ Answer/Explanation
Some acid would remain unreacted.
The final solution would be acidic.
The salt crystals would be contaminated.
Example 3 (Hard):
Describe fully how you would prepare a pure, dry sample of zinc sulfate using zinc oxide and sulfuric acid.
▶️ Answer/Explanation
Measure a fixed volume of dilute sulfuric acid into a beaker.
Heat the acid gently.
Add zinc oxide in small portions while stirring.
Continue adding until excess zinc oxide remains.
Filter to remove unreacted zinc oxide.
Collect the zinc sulfate solution.
Heat the solution to evaporate some water.
Allow it to cool to form crystals.
Filter and dry the crystals between filter papers.
This produces a pure, dry sample of zinc sulfate.
2.40C Preparing a Pure, Dry Sample of a Soluble Salt from an Acid and an Alkali
When preparing a soluble salt from an acid and an alkali, both reactants are soluble.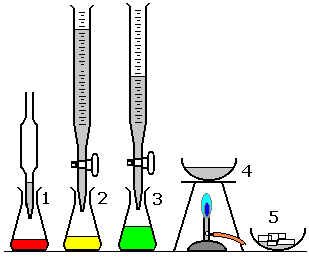
Therefore, excess reactant cannot be removed by filtration.
For this reason, a titration method must be used to obtain exact neutralisation.
Example Reaction
\( \mathrm{HCl(aq) + NaOH(aq) \rightarrow NaCl(aq) + H_2O(l)} \)
The aim is to obtain pure sodium chloride crystals.
Step 1: Perform a Titration
- Pipette a measured volume of alkali into a conical flask.
- Add a few drops of indicator (e.g. phenolphthalein).
- Fill the burette with the acid.
- Add acid slowly while swirling until the indicator just changes colour.
- Record the volume of acid used.
- Repeat to obtain concordant results.
This determines the exact volume of acid needed to neutralise the alkali.
Step 2: Prepare Pure Salt (Without Indicator)
- Repeat the reaction using the exact measured volumes.
- Do not add indicator.
- This ensures no contamination.
Step 3: Crystallisation
- Transfer the neutral solution to an evaporating basin.
- Heat gently to evaporate some water.
- Allow the solution to cool so crystals form.
- Filter the crystals.
- Dry between filter papers.
Why Titration Is Required
- Both reactants are soluble.
- Excess cannot be removed by filtration.
- Exact neutralisation ensures purity.
- Indicator is not added in final preparation to avoid contamination.
| Stage | Purpose |
|---|---|
| Titration | Find exact neutralisation point |
| Repeat without indicator | Obtain pure solution |
| Evaporation and crystallisation | Produce solid salt |
Example 1 (Conceptual):
Why must the salt be prepared again without the indicator?
▶️ Answer/Explanation
The indicator would contaminate the salt crystals.
To obtain a pure sample, no indicator is added in the final step.
Example 2 (Application):
Why can’t excess alkali be used and filtered off in this method?
▶️ Answer/Explanation
Alkalis are soluble in water.
They would remain dissolved in the solution.
Therefore filtration cannot remove the excess.
Example 3 (Hard ):
Describe fully how you would prepare a pure, dry sample of potassium sulfate from potassium hydroxide and sulfuric acid.
▶️ Answer/Explanation
Carry out a titration using potassium hydroxide in the conical flask and sulfuric acid in the burette.
Add indicator and determine the exact volume of acid needed for neutralisation.
Repeat the reaction using the exact measured volumes but without indicator.
The balanced equation is:
\( \mathrm{2KOH + H_2SO_4 \rightarrow K_2SO_4 + 2H_2O} \)
Transfer the neutral solution to an evaporating basin.
Heat gently to concentrate.
Allow to cool to form crystals.
Filter and dry the crystals to obtain pure potassium sulfate.
2.41C Preparing a Pure, Dry Sample of an Insoluble Salt (From Two Soluble Reactants)
An insoluble salt can be prepared by a precipitation reaction.
Two soluble salts are mixed together to form an insoluble solid called a precipitate.
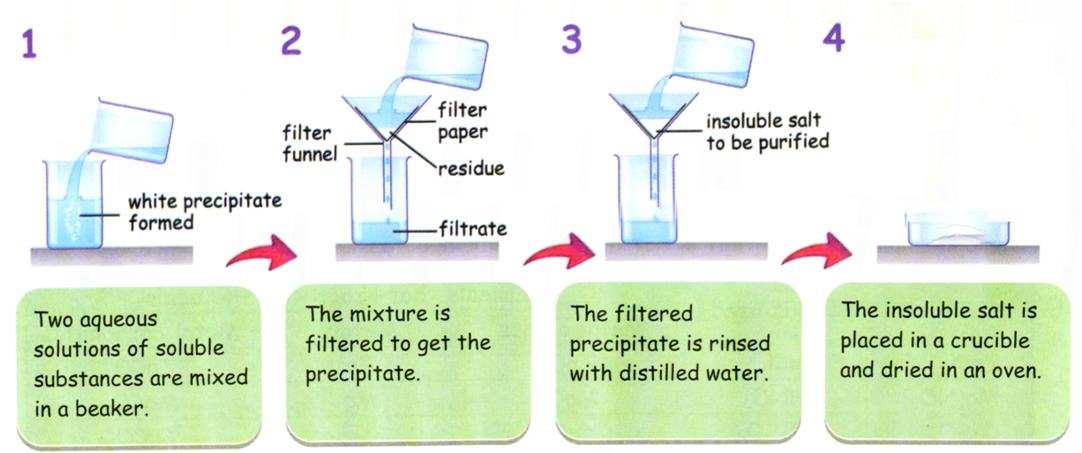
Example Reaction
Preparing barium sulfate from barium chloride and sodium sulfate:
\( \mathrm{BaCl_2(aq) + Na_2SO_4(aq) \rightarrow BaSO_4(s) + 2NaCl(aq)} \)
Barium sulfate is insoluble and forms a white precipitate.
The ionic equation is:
\( \mathrm{Ba^{2+}(aq) + SO_4^{2-}(aq) \rightarrow BaSO_4(s)} \)
Method
- Measure a known volume of one soluble salt solution into a beaker.
- Add the second soluble salt solution slowly while stirring.
- A precipitate will form.
- Filter the mixture using filter paper and funnel.
- Wash the precipitate with distilled water to remove soluble impurities.
- Dry the precipitate in a warm oven or between filter papers.
Why Washing Is Important
- The filtrate contains soluble by-products (e.g. sodium chloride).
- Washing removes these soluble impurities.
- This ensures the precipitate is pure.
| Stage | Purpose |
|---|---|
| Mix solutions | Form insoluble precipitate |
| Filtration | Separate solid from solution |
| Washing | Remove soluble impurities |
| Drying | Obtain pure, dry solid |
Key Idea
This method works because:
- The desired salt is insoluble.
- Other products remain dissolved in solution.
- The solid can be separated by filtration.
Example 1 (Conceptual):
Why must the precipitate be washed after filtration?
▶️ Answer/Explanation
To remove soluble impurities such as sodium chloride.
This ensures the salt obtained is pure.
Example 2 (Application):
What observation would you see when mixing barium chloride and sodium sulfate?
▶️ Answer/Explanation
A white precipitate forms.
This is insoluble barium sulfate.
Example 3 (Hard):
Describe fully how you would prepare a pure, dry sample of silver chloride from silver nitrate and sodium chloride.
▶️ Answer/Explanation
Measure silver nitrate solution into a beaker.
Add sodium chloride solution while stirring.
The reaction is:
\( \mathrm{AgNO_3(aq) + NaCl(aq) \rightarrow AgCl(s) + NaNO_3(aq)} \)
A white precipitate of silver chloride forms.
Filter to collect the solid.
Wash the precipitate with distilled water to remove sodium nitrate.
Dry the solid between filter papers or in a warm oven.
This produces a pure, dry sample of silver chloride.
