Edexcel iGCSE Chemistry -2.42 Practical: Copper(II) Sulfate Crystals- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -2.42 Practical: Copper(II) Sulfate Crystals- Study Notes- New syllabus
Edexcel iGCSE Chemistry -2.42 Practical: Copper(II) Sulfate Crystals- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
2.42 practical: prepare a sample of pure, dry hydrated copper(II) sulfate crystals starting from copper(II) oxide
2.42 Practical: Preparation of Hydrated Copper(II) Sulfate Crystals from Copper(II) Oxide
This experiment prepares a pure, dry sample of hydrated copper(II) sulfate crystals, \( \mathrm{CuSO_4 \cdot 5H_2O} \), starting from insoluble copper(II) oxide.
Principle of the Reaction
\( \mathrm{CuO(s) + H_2SO_4(aq) \rightarrow CuSO_4(aq) + H_2O(l)} \)
Copper(II) oxide is a base. Sulfuric acid is an acid. The reaction is neutralisation.
The blue crystals formed are hydrated copper(II) sulfate:
\( \mathrm{CuSO_4 \cdot 5H_2O} \)
Apparatus
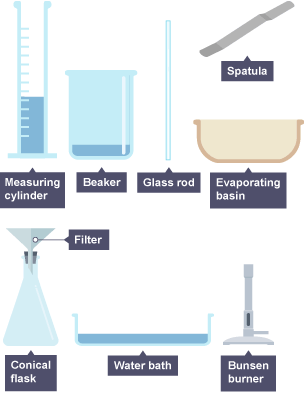
- Beaker
- Bunsen burner and tripod
- Glass stirring rod
- Filter funnel and filter paper
- Evaporating basin
- Measuring cylinder
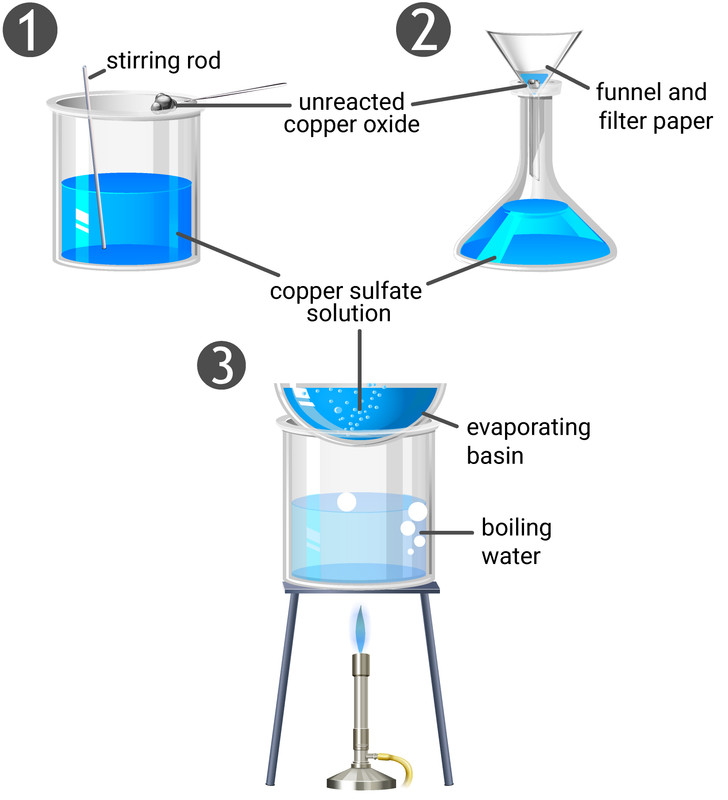
Method
- Measure a fixed volume of dilute sulfuric acid into a beaker.

- Heat the acid gently (do not boil).
- Add copper(II) oxide in small portions while stirring.
- Continue adding until some black solid remains unreacted (excess).
- Filter the mixture to remove excess copper(II) oxide.
- Collect the blue filtrate (copper(II) sulfate solution).
- Transfer the solution to an evaporating basin.
- Heat gently to evaporate some water until the solution becomes concentrated.
- Allow the solution to cool and crystallise.
- Filter the blue crystals.
- Dry the crystals between filter papers.
Observations
- Black copper(II) oxide is added.
- The solution gradually turns blue.
- Excess black solid remains at the bottom.
- After cooling, blue crystals form.
Why Each Step Is Important
| Step | Reason |
|---|---|
| Add excess CuO | Ensure all acid is neutralised |
| Filtration | Remove unreacted solid |
| Evaporation | Concentrate the solution |
| Cooling | Allow crystals to form |
Important Notes
- Do not evaporate to dryness , crystals may decompose.
- Hydrated crystals contain water of crystallisation.
- Overheating can drive off water of crystallisation.
Example 1 (Conceptual):
Why is excess copper(II) oxide added?
▶️ Answer/Explanation
To ensure all sulfuric acid is completely neutralised.
Excess solid can be removed by filtration.
Example 2 (Application):
Why must the solution be allowed to cool before crystals form?
▶️ Answer/Explanation
Solubility decreases as temperature decreases.
Cooling allows excess solute to crystallise out.
Example 3 (Hard – Full Explanation):
Explain fully how this method ensures the final product is pure hydrated copper(II) sulfate crystals.
▶️ Answer/Explanation
Excess copper(II) oxide ensures complete neutralisation of acid.
Filtration removes all unreacted solid.
The filtrate contains only copper(II) sulfate solution.
Evaporation concentrates the solution.
Cooling allows pure crystals to form.
Drying removes surface moisture without removing water of crystallisation.
Therefore the final product is pure hydrated \( \mathrm{CuSO_4 \cdot 5H_2O} \).