Edexcel iGCSE Chemistry -2.43C Practical: Lead(II) Sulfate- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -2.43C Practical: Lead(II) Sulfate- Study Notes- New syllabus
Edexcel iGCSE Chemistry -2.43C Practical: Lead(II) Sulfate- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
2.43C practical: prepare a sample of pure, dry lead(II) sulfate
2.43C Practical: Preparation of Pure, Dry Lead(II) Sulfate
Lead(II) sulfate, \( \mathrm{PbSO_4} \), is an insoluble salt.
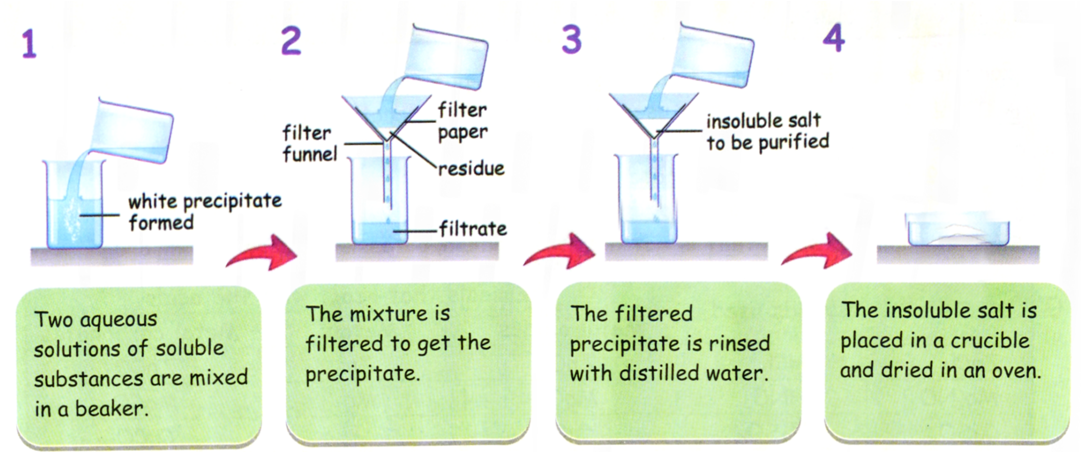
It is prepared using a precipitation reaction between two soluble salts.
Principle of the Reaction
A soluble lead(II) salt is reacted with a soluble sulfate salt.
Example:
\( \mathrm{Pb(NO_3)_2(aq) + Na_2SO_4(aq) \rightarrow PbSO_4(s) + 2NaNO_3(aq)} \)
The ionic equation is:
\( \mathrm{Pb^{2+}(aq) + SO_4^{2-}(aq) \rightarrow PbSO_4(s)} \)
Lead(II) sulfate forms as a white precipitate.
Apparatus
- Beakers
- Measuring cylinder
- Glass stirring rod
- Filter funnel and filter paper
- Distilled water
- Drying oven or filter paper
Method
- Measure a known volume of lead(II) nitrate solution into a beaker.
- Add sodium sulfate solution slowly while stirring.
- A white precipitate of lead(II) sulfate forms.
- Allow the precipitate to settle.
- Filter the mixture to collect the solid.
- Wash the precipitate with distilled water to remove soluble impurities (e.g. sodium nitrate).
- Dry the solid in a warm oven or between filter papers.
Observations
- Clear colourless solutions before mixing.
- White precipitate forms immediately.
- Solid remains on filter paper after filtration.
Why Washing Is Necessary
- The filtrate contains soluble sodium nitrate.
- Washing removes these impurities.
- This ensures the final solid is pure lead(II) sulfate.
Safety Considerations
- Lead compounds are toxic.
- Wear gloves and avoid ingestion.
- Dispose of waste properly.
| Stage | Purpose |
|---|---|
| Mix solutions | Form precipitate |
| Filtration | Separate solid |
| Washing | Remove soluble impurities |
| Drying | Obtain pure dry sample |
Example 1 (Conceptual):
Why can lead(II) sulfate be prepared by precipitation?
▶️ Answer/Explanation
Because lead(II) sulfate is insoluble in water.
It forms a solid when two soluble salts are mixed.
Example 2 (Application):
What would happen if the precipitate was not washed?
▶️ Answer/Explanation
Soluble impurities (e.g. sodium nitrate) would remain.
The final product would not be pure.
Example 3 (Hard):
Explain fully how this method ensures a pure, dry sample of lead(II) sulfate is obtained.
▶️ Answer/Explanation
Two soluble salts are reacted to form an insoluble precipitate of \( \mathrm{PbSO_4} \).
Filtration separates the solid from the solution.
Washing removes soluble by-products such as \( \mathrm{NaNO_3} \).
Drying removes water from the surface.
Therefore only pure, dry lead(II) sulfate remains.
