Edexcel iGCSE Chemistry -2.44 Tests for Gases- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -2.44 Tests for Gases- Study Notes- New syllabus
Edexcel iGCSE Chemistry -2.44 Tests for Gases- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
2.44 describe tests for these gases:
• hydrogen
• oxygen
• carbon dioxide
• ammonia
• chlorine
2.44 Tests for Common Gases
Certain gases can be identified using simple laboratory tests.
Each gas produces a characteristic observation.
1. Test for Hydrogen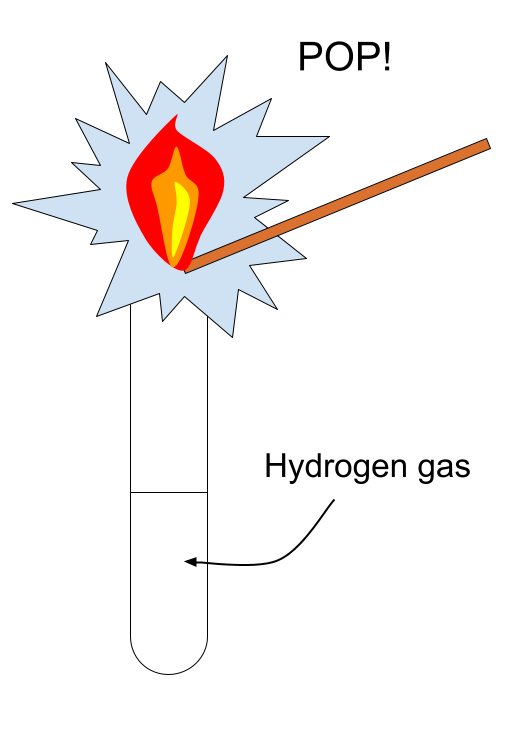
Procedure:
- Hold a lighted splint at the mouth of the test tube.
Positive result:
- A squeaky pop sound is heard.
Hydrogen combusts in oxygen:
\( \mathrm{2H_2(g) + O_2(g) \rightarrow 2H_2O(l)} \)
2. Test for Oxygen
Procedure: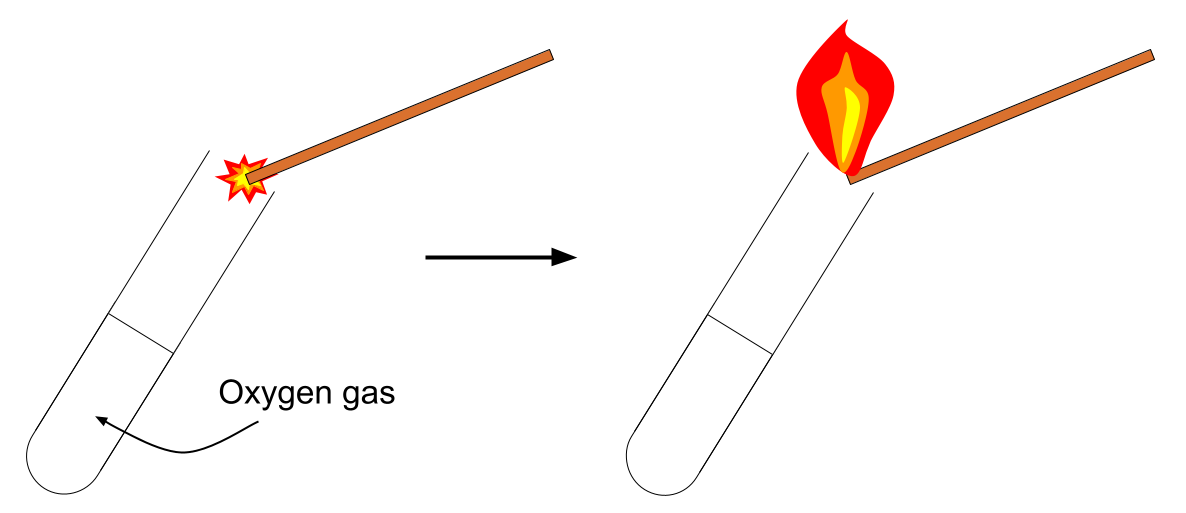
- Insert a glowing splint into the gas.
Positive result:
- The glowing splint relights.
Oxygen supports combustion.
3. Test for Carbon Dioxide
Procedure: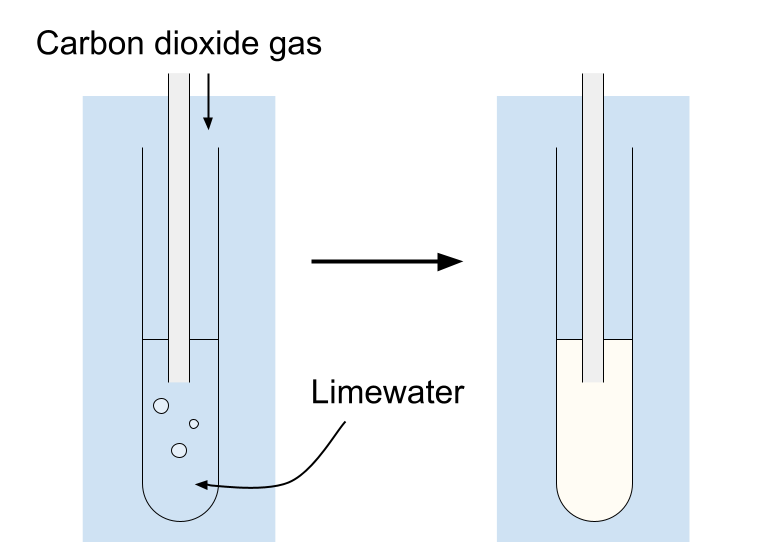
- Bubble the gas through limewater (calcium hydroxide solution).
Positive result:
- Limewater turns milky/cloudy.
Reaction:
\( \mathrm{Ca(OH)_2(aq) + CO_2(g) \rightarrow CaCO_3(s) + H_2O(l)} \)
Calcium carbonate forms as a white precipitate.
4. Test for Ammonia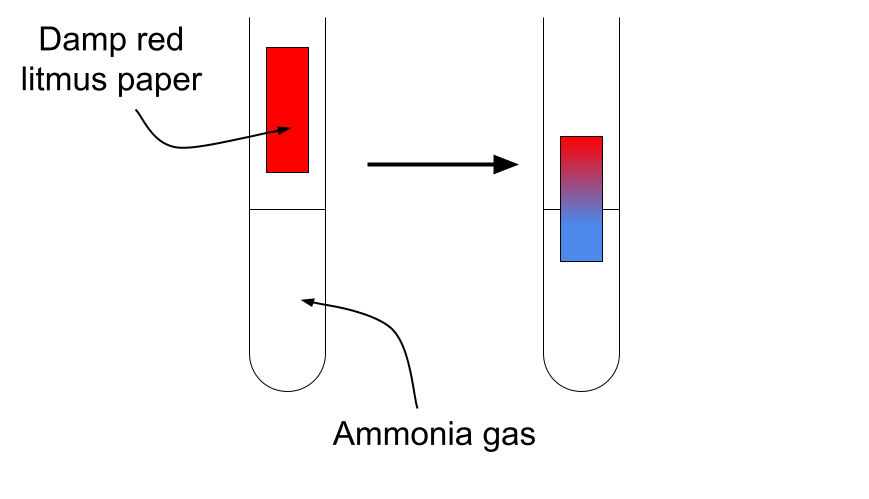
Procedure:
- Hold damp red litmus paper near the gas.
Positive result:
- Red litmus paper turns blue.
Ammonia is alkaline.
5. Test for Chlorine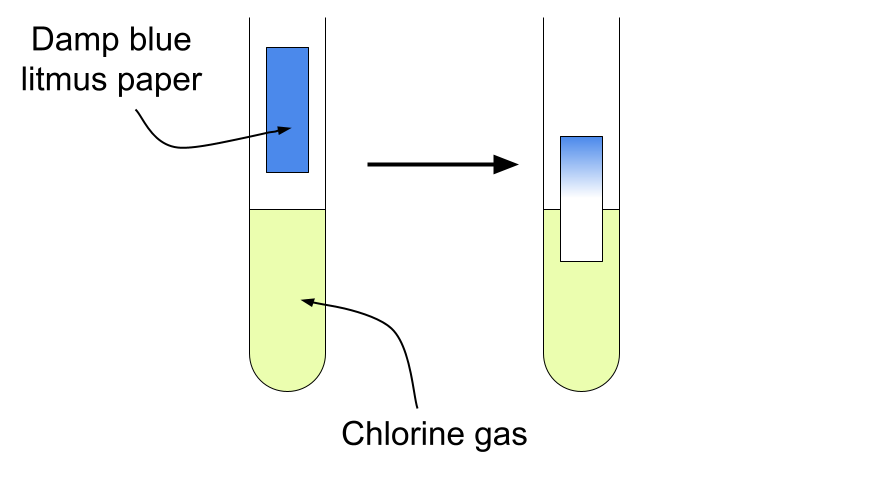
Procedure:
- Hold damp blue litmus paper in the gas.
Positive result:
- Litmus turns red and then is bleached white.
Chlorine is acidic and a bleaching agent.
| Gas | Test | Positive Result |
|---|---|---|
| Hydrogen | Lighted splint | Squeaky pop |
| Oxygen | Glowing splint | Relights |
| Carbon dioxide | Limewater | Turns milky |
| Ammonia | Damp red litmus | Turns blue |
| Chlorine | Damp blue litmus | Turns red then bleaches |
Example 1 (Conceptual):
Why must the litmus paper be damp when testing chlorine?
▶️ Answer/Explanation
Chlorine must dissolve in water first.
This forms acidic solution that affects the indicator.
Example 2 (Application):
A gas turns limewater milky. Identify the gas.
▶️ Answer/Explanation
The gas is carbon dioxide.
It forms calcium carbonate precipitate.
Example 3 (Hard):
A colourless gas gives a squeaky pop with a lighted splint. Explain fully what the gas is and why this occurs.
▶️ Answer/Explanation
The gas is hydrogen.
Hydrogen reacts rapidly with oxygen in the air.
The reaction is:
\( \mathrm{2H_2 + O_2 \rightarrow 2H_2O} \)
This rapid combustion produces the squeaky pop sound.
