Edexcel iGCSE Chemistry -2.45–2.46 Flame Tests and Flame Test Colours- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -2.45–2.46 Flame Tests and Flame Test Colours- Study Notes- New syllabus
Edexcel iGCSE Chemistry -2.45–2.46 Flame Tests and Flame Test Colours- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
2.45 describe how to carry out a flame test
2.46 know the colours formed in flame tests for these cations:
• Li⁺ is red
• Na⁺ is yellow
• K⁺ is lilac
• Ca²⁺ is orange-red
• Cu²⁺ is blue-green
2.45 Flame Tests
A flame test is used to identify certain metal ions based on the colour they produce in a Bunsen burner flame.
When heated, electrons in metal ions become excited. As the electrons return to lower energy levels, they release energy as coloured light.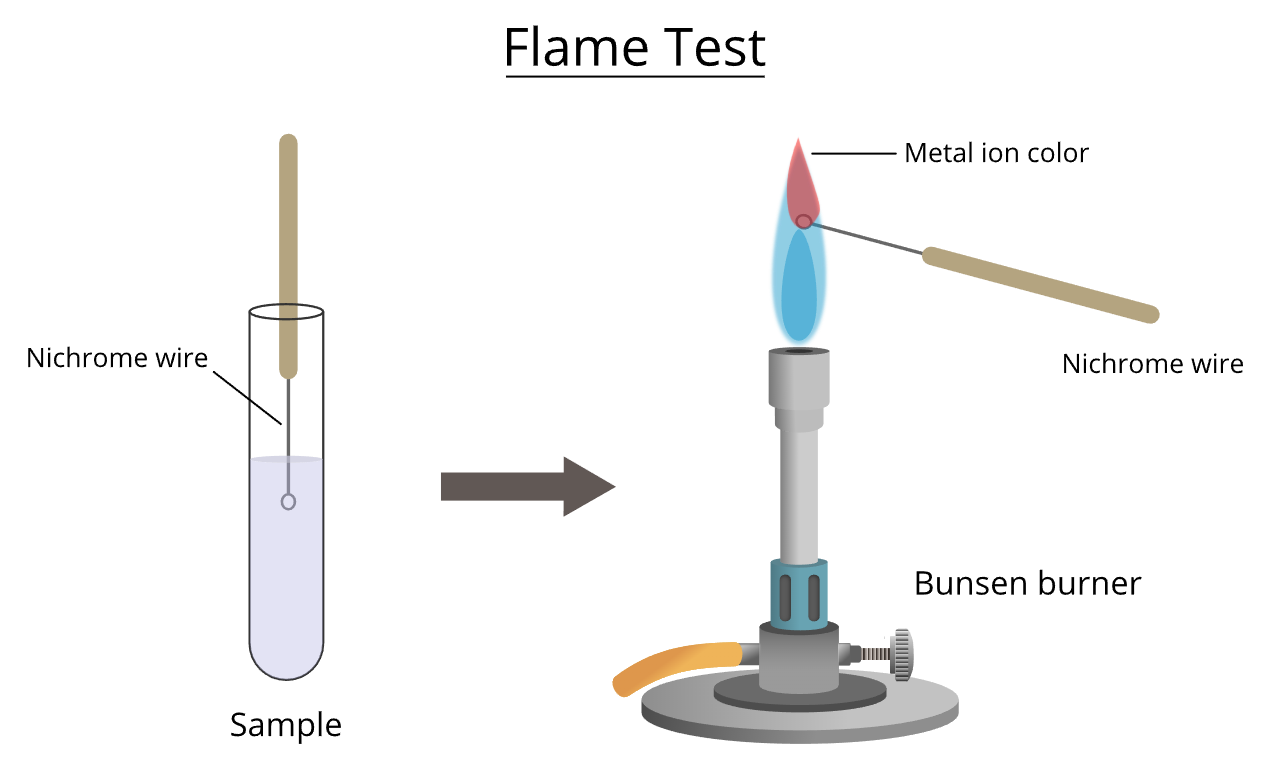
Apparatus
- Bunsen burner (blue flame)
- Platinum or nichrome wire loop
- Concentrated hydrochloric acid
- Sample of the compound
Method
- Dip the wire loop into concentrated hydrochloric acid.
- Heat the loop in the blue Bunsen flame until no colour is seen (to clean it).
- Dip the clean loop into the sample.
- Place the loop in the edge of the blue flame.
- Observe the colour of the flame.
Flame Colours of Common Metal Ions
| Metal Ion | Flame Colour |
|---|---|
| Lithium \( \mathrm{Li^+} \) | Crimson red |
| Sodium \( \mathrm{Na^+} \) | Yellow |
| Potassium \( \mathrm{K^+} \) | Lilac |
| Calcium \( \mathrm{Ca^{2+}} \) | Orange-red |
| Copper(II) \( \mathrm{Cu^{2+}} \) | Blue-green |
Important Notes
- The wire must be cleaned before each test.
- Use a blue (non-luminous) Bunsen flame.
- Sodium contamination can mask other colours (very strong yellow).
Example 1 (Conceptual):
Why must the wire loop be cleaned before testing a new sample?
▶️ Answer/Explanation
To remove any previous metal ions.
This prevents contamination and incorrect colours.
Example 2 (Application):
A compound produces a lilac flame. Which metal ion is present?
▶️ Answer/Explanation
The lilac colour indicates potassium ions \( \mathrm{K^+} \).
Example 3 (Hard):
A flame test gives a strong yellow colour, even though the compound is believed to contain potassium. Explain fully.
▶️ Answer/Explanation
Sodium ions produce an intense yellow flame.
Even small amounts of sodium contamination can mask other colours.
The strong yellow suggests sodium is present.
The potassium lilac colour may be hidden.
2.46 Colours Formed in Flame Tests for Specific Cations
Certain metal cations produce characteristic colours when heated in a Bunsen flame.

The colour is caused by excited electrons releasing energy as visible light.
Required Flame Test Colours
| Cation | Flame Colour |
|---|---|
| \( \mathrm{Li^+} \) | Red (crimson) |
| \( \mathrm{Na^+} \) | Yellow |
| \( \mathrm{K^+} \) | Lilac |
| \( \mathrm{Ca^{2+}} \) | Orange-red |
| \( \mathrm{Cu^{2+}} \) | Blue-green |
Important Exam Notes
- Sodium produces a very strong yellow flame.
- Small traces of sodium can mask other colours.
- Use a clean wire loop to avoid contamination.
Quick Memory Pattern
- Lithium → Red
- Sodium → Yellow
- Potassium → Lilac
- Calcium → Orange-red
- Copper(II) → Blue-green
Example 1 (Conceptual):
Which ion produces a blue-green flame?
▶️ Answer/Explanation
\( \mathrm{Cu^{2+}} \)
Example 2 (Application):
A compound gives a yellow flame. Which ion is present?
▶️ Answer/Explanation
\( \mathrm{Na^+} \)
Example 3 (Hard):
A sample gives an orange-red flame. Explain fully which ion is present and why contamination might affect the result.
▶️ Answer/Explanation
An orange-red flame indicates \( \mathrm{Ca^{2+}} \).
However, if sodium is present as a contaminant, it may produce a strong yellow flame.
This yellow can mask the calcium colour.
Therefore the wire must be cleaned carefully before testing.
