Edexcel iGCSE Chemistry -2.47 Tests for Cations- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -2.47 Tests for Cations- Study Notes- New syllabus
Edexcel iGCSE Chemistry -2.47 Tests for Cations- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
2.47 describe tests for these cations:
• NH₄⁺ using sodium hydroxide solution
• Cu²⁺, Fe²⁺ and Fe³⁺ using sodium hydroxide solution
2.47 Tests for Cations Using Sodium Hydroxide Solution
Certain positive ions (cations) can be identified by adding sodium hydroxide solution.
Observations include formation of precipitates or production of a gas.
1. Test for Ammonium Ion, \( \mathrm{NH_4^+} \) 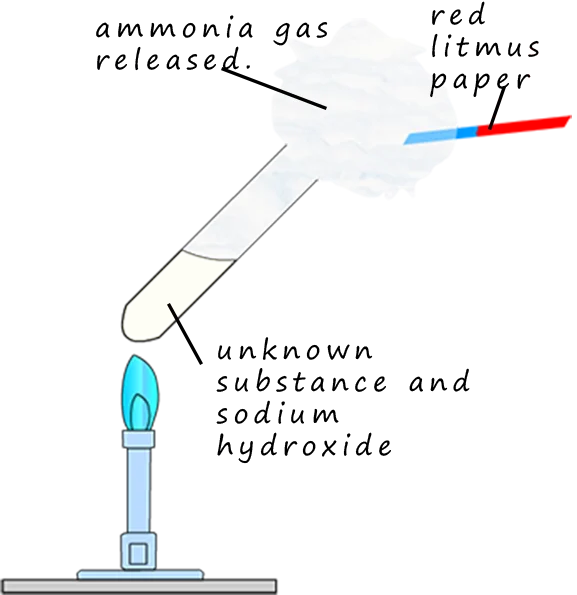
Procedure:
- Add sodium hydroxide solution to the sample.
- Warm the mixture gently.
- Hold damp red litmus paper near the mouth of the test tube.
Reaction:
\( \mathrm{NH_4^+(aq) + OH^-(aq) \rightarrow NH_3(g) + H_2O(l)} \)
Observation:
- Pungent gas produced (ammonia).
- Damp red litmus turns blue.
This confirms the presence of \( \mathrm{NH_4^+} \).
2. Test for Copper(II) Ion, \( \mathrm{Cu^{2+}} \)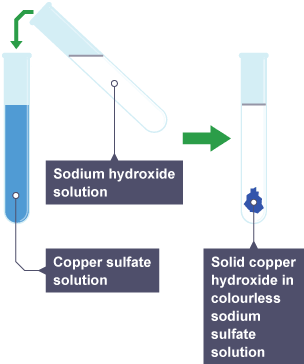
Procedure:
- Add sodium hydroxide solution to the sample.
Reaction:
\( \mathrm{Cu^{2+}(aq) + 2OH^-(aq) \rightarrow Cu(OH)_2(s)} \)
Observation:
- Blue precipitate forms.
- Insoluble in excess sodium hydroxide.
3. Test for Iron(II) Ion, \( \mathrm{Fe^{2+}} \)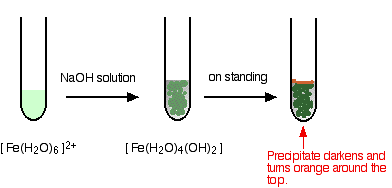
\( \mathrm{Fe^{2+}(aq) + 2OH^-(aq) \rightarrow Fe(OH)_2(s)} \)
Observation:
- Green precipitate forms.
- Slowly turns brown on standing (due to oxidation).
4. Test for Iron(III) Ion, \( \mathrm{Fe^{3+}} \)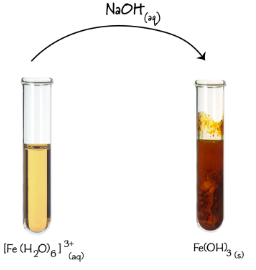
\( \mathrm{Fe^{3+}(aq) + 3OH^-(aq) \rightarrow Fe(OH)_3(s)} \)
Observation:
- Brown precipitate forms.
- Insoluble in excess sodium hydroxide.
| Cation | Observation with NaOH |
|---|---|
| \( \mathrm{NH_4^+} \) | Ammonia gas evolved, turns damp red litmus blue |
| \( \mathrm{Cu^{2+}} \) | Blue precipitate |
| \( \mathrm{Fe^{2+}} \) | Green precipitate (turns brown) |
| \( \mathrm{Fe^{3+}} \) | Brown precipitate |
Example 1 (Conceptual):
Why must the ammonium test be warmed?
▶️ Answer/Explanation
Warming helps release ammonia gas.
This makes the gas easier to detect.
Example 2 (Application):
A blue precipitate forms when sodium hydroxide is added. Which ion is present?
▶️ Answer/Explanation
\( \mathrm{Cu^{2+}} \) is present.
It forms blue \( \mathrm{Cu(OH)_2} \).
Example 3 (Hard):
A green precipitate forms and slowly turns brown when left exposed to air. Identify the ion and explain fully.
▶️ Answer/Explanation
The green precipitate indicates \( \mathrm{Fe^{2+}} \).
The reaction is:
\( \mathrm{Fe^{2+} + 2OH^- \rightarrow Fe(OH)_2} \)
Iron(II) hydroxide slowly oxidises in air to iron(III) hydroxide.
Iron(III) hydroxide is brown.
This colour change confirms \( \mathrm{Fe^{2+}} \) was present.
