Edexcel iGCSE Chemistry -2.48 Tests for Anions- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -2.48 Tests for Anions- Study Notes- New syllabus
Edexcel iGCSE Chemistry -2.48 Tests for Anions- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
2.48 describe tests for these anions:
• Cl⁻, Br⁻ and I⁻ using acidified silver nitrate solution
• SO₄²⁻ using acidified barium chloride solution
• CO₃²⁻ using hydrochloric acid
2.48 Tests for Common Anions
Certain negative ions (anions) can be identified using characteristic chemical tests.
The tests involve forming a precipitate or detecting a gas.
1. Tests for Halide Ions: \( \mathrm{Cl^-} \), \( \mathrm{Br^-} \), \( \mathrm{I^-} \)
Reagent: Acidified silver nitrate solution
Procedure:
- Add a few drops of dilute nitric acid to the sample.
- Add silver nitrate solution.
Nitric acid is used to remove carbonate ions that may interfere.
Reactions:
\( \mathrm{Ag^+(aq) + Cl^-(aq) \rightarrow AgCl(s)} \)
\( \mathrm{Ag^+(aq) + Br^-(aq) \rightarrow AgBr(s)} \)
\( \mathrm{Ag^+(aq) + I^-(aq) \rightarrow AgI(s)} \)
Observations:
| Ion | Precipitate Colour |
|---|---|
| \( \mathrm{Cl^-} \) | White |
| \( \mathrm{Br^-} \) | Cream |
| \( \mathrm{I^-} \) | Yellow |
2. Test for Sulfate Ion, \( \mathrm{SO_4^{2-}} \)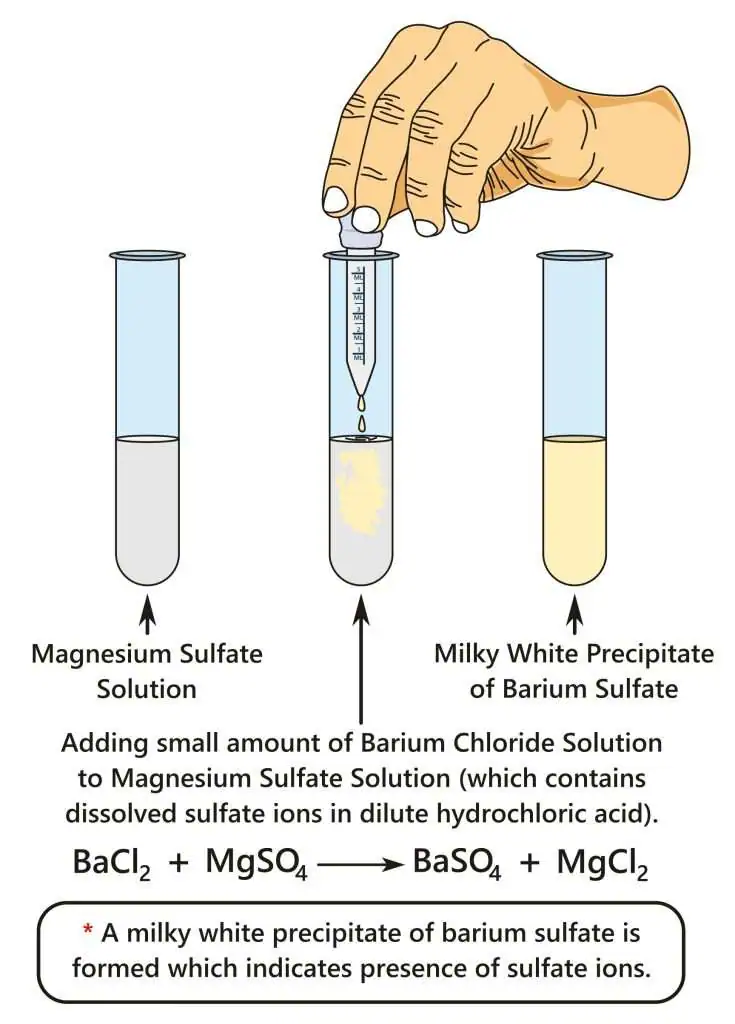
Reagent: Acidified barium chloride solution
Procedure:
- Add dilute hydrochloric acid to the sample.
- Add barium chloride solution.
Reaction:
\( \mathrm{Ba^{2+}(aq) + SO_4^{2-}(aq) \rightarrow BaSO_4(s)} \)
Observation:
- White precipitate forms.
- Precipitate is insoluble in acid.
3. Test for Carbonate Ion, \( \mathrm{CO_3^{2-}} \)
Reagent: Dilute hydrochloric acid
Procedure:
- Add dilute hydrochloric acid to the sample.
- Test the gas produced using limewater.
Reaction:
\( \mathrm{CO_3^{2-}(aq) + 2H^+(aq) \rightarrow CO_2(g) + H_2O(l)} \)
Observation:
- Effervescence (bubbling).
- The gas turns limewater milky.
\( \mathrm{Ca(OH)_2(aq) + CO_2(g) \rightarrow CaCO_3(s) + H_2O(l)} \)
| Anion | Test | Positive Result |
|---|---|---|
| \( \mathrm{Cl^-} \) | Acidified \( \mathrm{AgNO_3} \) | White precipitate |
| \( \mathrm{Br^-} \) | Acidified \( \mathrm{AgNO_3} \) | Cream precipitate |
| \( \mathrm{I^-} \) | Acidified \( \mathrm{AgNO_3} \) | Yellow precipitate |
| \( \mathrm{SO_4^{2-}} \) | Acidified \( \mathrm{BaCl_2} \) | White precipitate |
| \( \mathrm{CO_3^{2-}} \) | Dilute HCl + limewater | Bubbling, limewater milky |
Example 1 (Conceptual):
Why is nitric acid added before silver nitrate in the halide test?
▶️ Answer/Explanation
Nitric acid removes carbonate ions.
This prevents a false positive white precipitate.
Example 2 (Application):
A cream precipitate forms when acidified silver nitrate is added. Which ion is present?
▶️ Answer/Explanation
A cream precipitate indicates \( \mathrm{Br^-} \).
Example 3 (Hard):
A sample produces effervescence when hydrochloric acid is added and the gas turns limewater milky. Identify the ion and explain fully.
▶️ Answer/Explanation
Effervescence shows a gas is produced.
The gas turns limewater milky, so it is carbon dioxide.
This means carbonate ions were present.
The reaction is:
\( \mathrm{CO_3^{2-} + 2H^+ \rightarrow CO_2 + H_2O} \)
Therefore \( \mathrm{CO_3^{2-}} \) is present.
