Edexcel iGCSE Chemistry -2.49–2.50 Tests for Water and Pure Water- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -2.49–2.50 Tests for Water and Pure Water- Study Notes- New syllabus
Edexcel iGCSE Chemistry -2.49–2.50 Tests for Water and Pure Water- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
2.49 describe a test for the presence of water using anhydrous copper(II) sulfate
2.50 describe a physical test to show whether a sample of water is pure
2.49 Test for the Presence of Water Using Anhydrous Copper(II) Sulfate
Water can be identified using anhydrous copper(II) sulfate.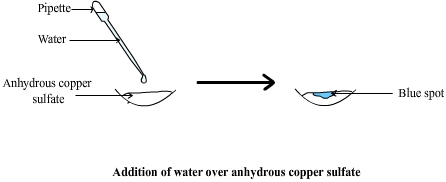
“Anhydrous” means without water.
Anhydrous copper(II) sulfate is white.
When water is present, it becomes hydrated and turns blue.
Chemical Reaction
\( \mathrm{CuSO_4(s) + 5H_2O(l) \rightarrow CuSO_4 \cdot 5H_2O(s)} \)
White anhydrous copper(II) sulfate reacts with water to form blue hydrated copper(II) sulfate.
Procedure
- Place a small amount of anhydrous copper(II) sulfate in a test tube or on a watch glass.
- Add a few drops of the liquid being tested (or expose the solid to the vapour).
- Observe any colour change.
Positive Result
- White solid turns blue.
This confirms the presence of water.
Important Notes
- The test only confirms the presence of water.
- It does not measure how much water is present.
- Do not confuse hydrated (blue) and anhydrous (white) forms.
| Substance | Colour |
|---|---|
| Anhydrous \( \mathrm{CuSO_4} \) | White |
| Hydrated \( \mathrm{CuSO_4 \cdot 5H_2O} \) | Blue |
Example 1 (Conceptual):
Why does anhydrous copper(II) sulfate turn blue when water is added?
▶️ Answer/Explanation
Water molecules attach to the copper(II) sulfate.
This forms hydrated copper(II) sulfate.
The hydrated form is blue in colour.
Example 2 (Application):
A white solid turns blue when exposed to steam. What does this show?
▶️ Answer/Explanation
Water vapour is present.
The white anhydrous copper(II) sulfate reacted with water.
Example 3 (Hard):
A student heats blue copper(II) sulfate until it turns white. Explain fully what has happened.
▶️ Answer/Explanation
Blue copper(II) sulfate contains water of crystallisation:
\( \mathrm{CuSO_4 \cdot 5H_2O} \)
Heating removes this water:
\( \mathrm{CuSO_4 \cdot 5H_2O \rightarrow CuSO_4 + 5H_2O} \)
The solid becomes white anhydrous copper(II) sulfate.
The colour change shows water has been removed.
2.50 Physical Test to Determine if a Sample of Water is Pure
A pure substance has a fixed melting point and a fixed boiling point.
Therefore, the purity of water can be tested by measuring its boiling point or melting point.
Boiling Point Test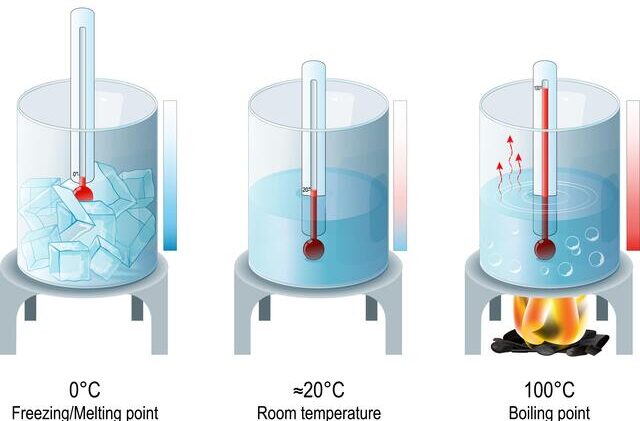
- Place the water sample in a beaker.
- Insert a thermometer.
- Heat the water.
- Record the temperature at which it boils.
Pure water boils at:
\( 100^\circ \mathrm{C} \) (at standard atmospheric pressure)
If the boiling point is higher than \( 100^\circ \mathrm{C} \), impurities are present.
Melting (Freezing) Point Test
- Cool the water sample.
- Measure the temperature at which it freezes.
Pure water freezes at:
\( 0^\circ \mathrm{C} \)
If it freezes below \( 0^\circ \mathrm{C} \), impurities are present.
Why This Works
Impurities:
- Increase boiling point
- Lower freezing point
- Cause temperature ranges instead of fixed points
| Property | Pure Water | Impure Water |
|---|---|---|
| Boiling point | \( 100^\circ \mathrm{C} \) | Above \( 100^\circ \mathrm{C} \) |
| Freezing point | \( 0^\circ \mathrm{C} \) | Below \( 0^\circ \mathrm{C} \) |
Important Exam Note
For GCSE answers:
- State the expected temperature value.
- Mention “fixed temperature” if the sample is pure.
- Mention “range” if impure.
Example 1 (Conceptual):
Why does impure water boil at a higher temperature?
▶️ Answer/Explanation
Dissolved particles interfere with evaporation.
More energy is required to boil the liquid.
Example 2 (Application):
A water sample boils at \( 101.5^\circ \mathrm{C} \). What does this show?
▶️ Answer/Explanation
The water is not pure.
Impurities have increased the boiling point.
Example 3 (Hard):
A student measures the freezing point of a water sample and finds it freezes gradually between \( -1^\circ \mathrm{C} \) and \( 0^\circ \mathrm{C} \). Explain fully what this shows.
▶️ Answer/Explanation
A pure substance freezes at a fixed temperature.
The sample freezes over a range of temperatures.
This indicates it is a mixture.
The freezing point has been lowered by impurities.
Therefore the water sample is impure.
