Edexcel iGCSE Chemistry -2.4C Reactivity Trend and Electronic Configuration- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -2.4C Reactivity Trend and Electronic Configuration- Study Notes- New syllabus
Edexcel iGCSE Chemistry -2.4C Reactivity Trend and Electronic Configuration- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
2.4C explain the trend in reactivity in Group 1 in terms of electronic configurations
2.4C Explaining the Trend in Reactivity in Group 1 Using Electronic Configurations
Group 1 elements are called the alkali metals. They include lithium, sodium, potassium, rubidium and caesium.
Reactivity increases down Group 1. This trend can be explained using electronic configurations.
Electronic Structure of Group 1 Elements
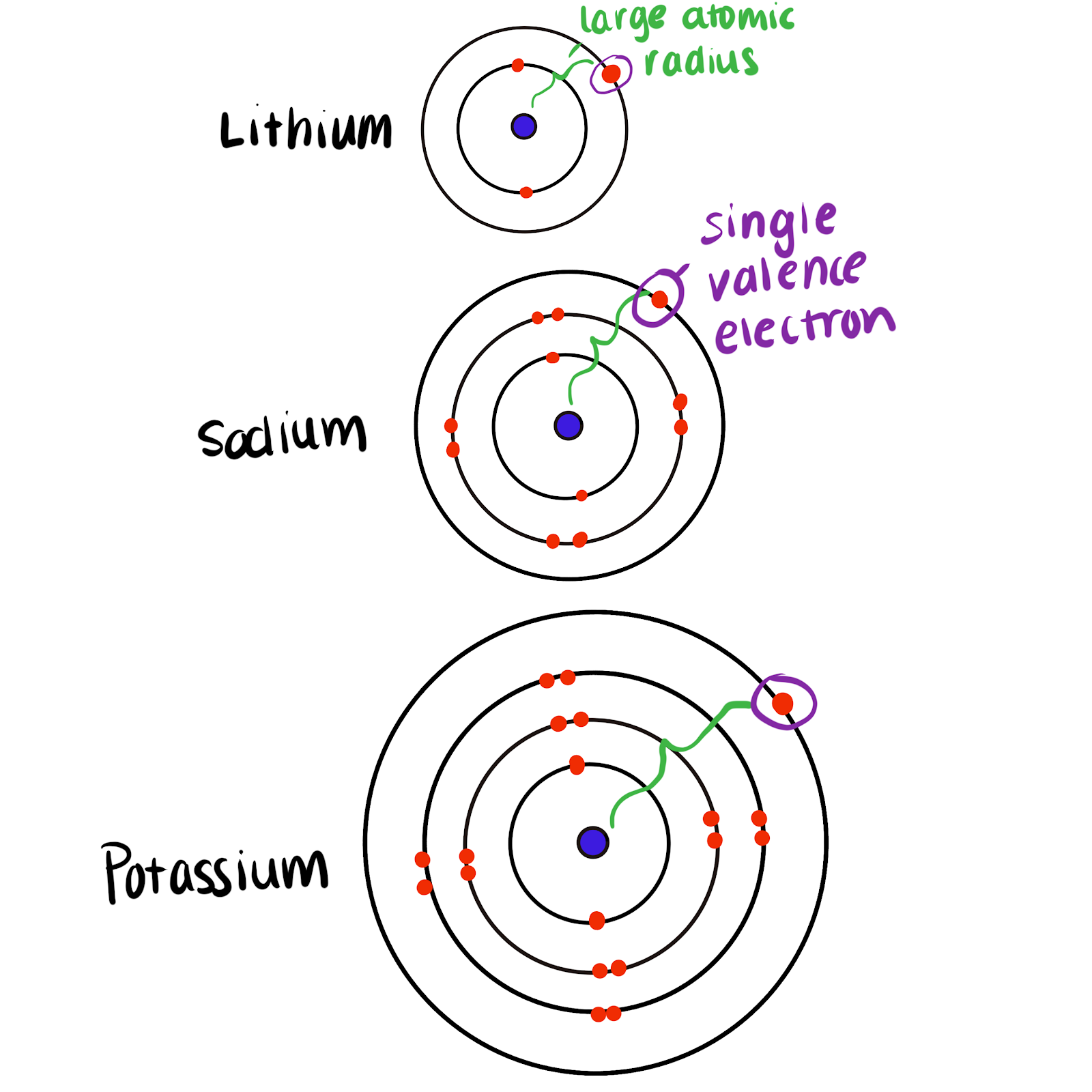
- Lithium: \( 2,1 \)
- Sodium: \( 2,8,1 \)
- Potassium: \( 2,8,8,1 \)
All Group 1 elements have one electron in their outer shell.
How They React
Group 1 metals react by losing their outer electron to form a positive ion:
\( \mathrm{M \rightarrow M^+ + e^-} \)
This gives them a full outer shell.
Why Reactivity Increases Down the Group
- Down the group, the number of electron shells increases.
- The outer electron is further from the nucleus.
- There is increased shielding from inner shells.
- The attraction between the nucleus and the outer electron decreases.
- The outer electron is lost more easily.
- Therefore reactivity increases.
| Element | Electronic Configuration | Distance of Outer Electron | Reactivity |
|---|---|---|---|
| Lithium | \( 2,1 \) | Closest | Lowest |
| Sodium | \( 2,8,1 \) | Further | Higher |
| Potassium | \( 2,8,8,1 \) | Furthest | Highest |
Example 1 (Conceptual):
Why do all Group 1 elements have similar chemical properties?
▶️ Answer/Explanation
They all have one outer electron.
They react by losing this electron.
This gives them similar chemical behaviour.
Example 2 (Application):
Which is more reactive, sodium or potassium? Explain using electronic structure.
▶️ Answer/Explanation
Potassium is more reactive.
It has more electron shells.
The outer electron is further from the nucleus.
It is lost more easily.
Example 3 (Hard):
Explain fully why reactivity increases down Group 1 using electronic configurations and shielding.
▶️ Answer/Explanation
All Group 1 elements have one outer shell electron.
Down the group, atoms have more electron shells.
The outer electron is further from the nucleus.
Inner shells cause shielding.
The attraction between the nucleus and outer electron decreases.
The outer electron is lost more easily.
Therefore reactivity increases down the group.
