Edexcel iGCSE Chemistry -2.5 Physical Properties of Halogens- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -2.5 Physical Properties of Halogens- Study Notes- New syllabus
Edexcel iGCSE Chemistry -2.5 Physical Properties of Halogens- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
2.5 know the colours, physical states (at room temperature) and trends in physical properties of these elements
2.5 Colours, Physical States and Trends in Physical Properties of Group 1 Elements
Group 1 elements (alkali metals) include lithium, sodium and potassium.
They share similar physical properties because they have one outer electron and similar metallic structures.
Colours and Physical States (Room Temperature)
- Lithium – silvery-grey solid
- Sodium – silvery-grey solid
- Potassium – silvery-grey solid
All are soft metals that can be cut with a knife.
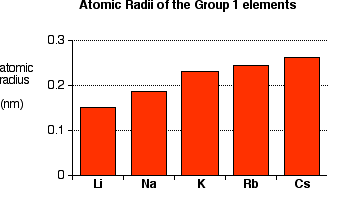
Trends in Physical Properties Down Group 1
- Melting point decreases.
- Boiling point decreases.
- Density generally increases.
- Softness increases.
- Atomic radius increases.
Melting Point Trend
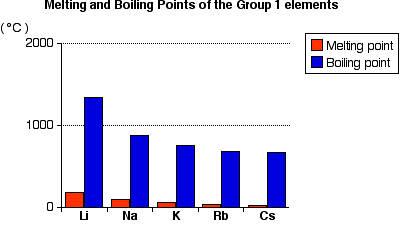
- Metallic bonding becomes weaker down the group.
- Atoms become larger.
- The attraction between positive ions and delocalised electrons weakens.
- Less energy is needed to melt the metal.
Density Trend
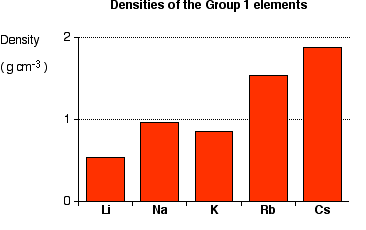
- Generally increases down the group.
- Atoms have greater mass.
- However, all Group 1 metals have relatively low densities.
- Lithium, sodium and potassium float on water.
| Element | Colour | State at Room Temp | Melting Point Trend |
|---|---|---|---|
| Lithium | Silvery-grey | Solid | Highest (among the three) |
| Sodium | Silvery-grey | Solid | Lower |
| Potassium | Silvery-grey | Solid | Lowest |
Example 1 (Conceptual):
Why do melting points decrease down Group 1?
▶️ Answer/Explanation
Atomic size increases down the group.
Metallic bonding becomes weaker.
Less energy is needed to break the bonds.
Therefore melting point decreases.
Example 2 (Application):
Predict whether rubidium would have a higher or lower melting point than potassium.
▶️ Answer/Explanation
Lower melting point.
Melting point decreases down the group.
Example 3 (Hard):
Explain fully why Group 1 metals are soft and why their melting points decrease down the group.
▶️ Answer/Explanation
Group 1 metals have one delocalised electron per atom.
This forms metallic bonds between positive ions and delocalised electrons.
Because there is only one outer electron, the metallic bonding is relatively weak.
This makes the metals soft.
Down the group, atomic radius increases.
The positive ions become larger.
The attraction between ions and delocalised electrons weakens.
Therefore less energy is needed to melt the metal.
Melting points decrease down the group.
