Edexcel iGCSE Chemistry -2.6 Trends in Group 7- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -2.6 Trends in Group 7- Study Notes- New syllabus
Edexcel iGCSE Chemistry -2.6 Trends in Group 7- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
2.6 use knowledge of trends in Group 7 to predict the properties of other halogens
2.6 Using Trends in Group 7 to Predict the Properties of Other Halogens
Group 7 elements are called the halogens. They include:
- Fluorine (F)
- Chlorine (Cl)
- Bromine (Br)
- Iodine (I)
By understanding the trends down Group 7, we can predict the properties of halogens that are not directly studied (e.g. astatine).
Electronic Structure of Halogens
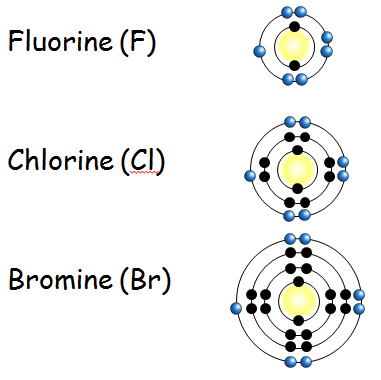
- All halogens have 7 electrons in their outer shell.
- They react by gaining 1 electron to form a negative ion.
\( \mathrm{X + e^- \rightarrow X^-} \)
Key Trends Down Group 7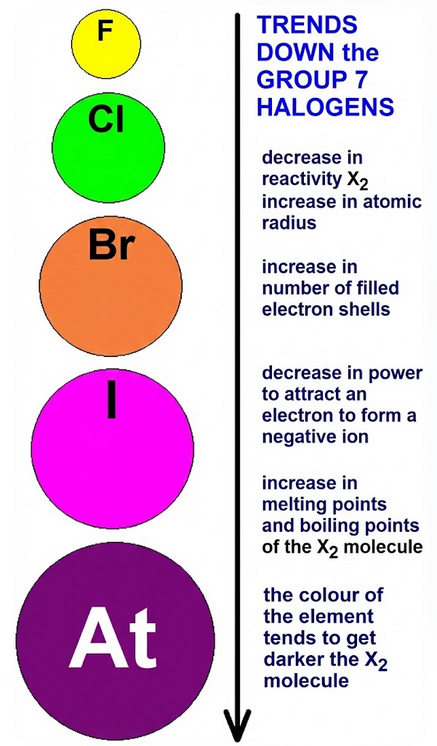
- Reactivity decreases.
- Melting point increases.
- Boiling point increases.
- Density increases.
- Colour becomes darker.
Reactivity Trend
- Halogens react by gaining an electron.
- Down the group, atomic size increases.
- The outer shell is further from the nucleus.
- There is more shielding by inner electrons.
- The attraction for an extra electron decreases.
- Reactivity decreases down the group.
Therefore iodine is less reactive than chlorine.
Physical State Trend (Room Temperature)
- Fluorine – pale yellow gas.
- Chlorine – green gas.
- Bromine – red-brown liquid.
- Iodine – grey solid.
Down the group, intermolecular forces become stronger because molecules become larger.
| Property | Trend Down Group 7 | Prediction for Astatine |
|---|---|---|
| Reactivity | Decreases | Very low |
| Melting point | Increases | Higher than iodine |
| State | Gas → liquid → solid | Solid |
| Colour | Darker | Very dark solid |
Example 1 (Conceptual):
Why does reactivity decrease down Group 7?
▶️ Answer/Explanation
Atomic radius increases.
Shielding increases.
Attraction for an extra electron decreases.
Therefore reactivity decreases.
Example 2 (Application):
Predict the physical state of astatine at room temperature.
▶️ Answer/Explanation
It would be a solid.
Melting and boiling points increase down the group.
Example 3 (Hard):
Explain fully how knowledge of trends in Group 7 allows prediction of the properties of astatine.
▶️ Answer/Explanation
Halogens have seven outer electrons and react by gaining one electron.
Down the group, atomic size increases and shielding increases.
The attraction for an incoming electron decreases, so reactivity decreases.
Intermolecular forces become stronger as molecular size increases.
Therefore melting and boiling points increase.
Since iodine is a solid, astatine (below iodine) would also be a solid with an even higher melting point and lower reactivity.
