Edexcel iGCSE Chemistry -2.7 Displacement Reactions- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -2.7 Displacement Reactions- Study Notes- New syllabus
Edexcel iGCSE Chemistry -2.7 Displacement Reactions- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
2.7 understand how displacement reactions involving halogens and halides provide evidence for the trend in reactivity in Group 7
2.7 Displacement Reactions as Evidence for the Reactivity Trend in Group 7
Displacement reactions between halogens and halide ions provide clear evidence for the trend in reactivity down Group 7.
A more reactive halogen will displace a less reactive halogen from its compound.
Reactivity Trend in Group 7
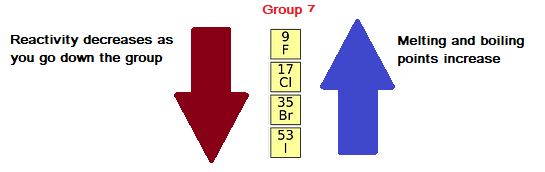
- Reactivity decreases down the group.
- Chlorine is more reactive than bromine.
- Bromine is more reactive than iodine.
This is because halogens react by gaining one electron.
- Down the group, atomic size increases.
- Shielding increases.
- The attraction for an extra electron decreases.
- Therefore reactivity decreases.
General Rule for Displacement
A more reactive halogen will displace a less reactive halide ion from solution.
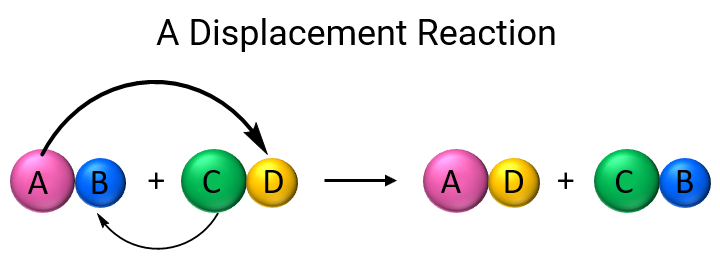
Examples of Displacement Reactions
Chlorine and Potassium Bromide:
\( \mathrm{Cl_2(aq) + 2KBr(aq) \rightarrow 2KCl(aq) + Br_2(aq)} \)
Chlorine displaces bromine because chlorine is more reactive.
Chlorine and Potassium Iodide:
\( \mathrm{Cl_2(aq) + 2KI(aq) \rightarrow 2KCl(aq) + I_2(aq)} \)
Chlorine displaces iodine.
Bromine and Potassium Iodide:
\( \mathrm{Br_2(aq) + 2KI(aq) \rightarrow 2KBr(aq) + I_2(aq)} \)
Bromine displaces iodine.
Iodine and Potassium Chloride:
No reaction occurs because iodine is less reactive than chlorine.
| Halogen Added | Halide Solution | Reaction? | Conclusion |
|---|---|---|---|
| Chlorine | Bromide | Yes | Chlorine more reactive |
| Chlorine | Iodide | Yes | Chlorine more reactive |
| Bromine | Iodide | Yes | Bromine more reactive |
| Iodine | Chloride | No | Iodine less reactive |
Example 1 (Conceptual):
Why does chlorine displace bromine from potassium bromide?
▶️ Answer/Explanation
Chlorine is more reactive than bromine.
It gains electrons more easily.
It displaces bromine from its compound.
Example 2 (Application):
Predict what happens when bromine is added to potassium chloride.
▶️ Answer/Explanation
No reaction occurs.
Bromine is less reactive than chlorine.
It cannot displace chloride ions.
Example 3 (Hard):
Explain fully how displacement reactions provide evidence for the trend in reactivity down Group 7.
▶️ Answer/Explanation
Displacement reactions show that a more reactive halogen can displace a less reactive halide from solution.
Chlorine displaces bromine and iodine.
Bromine displaces iodine.
Iodine does not displace chlorine or bromine.
This shows that reactivity decreases down the group.
The trend is explained by increasing atomic size and shielding down the group.
The attraction for an extra electron decreases.
Therefore reactivity decreases.
