Edexcel iGCSE Chemistry -2.8C Reactivity Trend and Electronic Configuration- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -2.8C Reactivity Trend and Electronic Configuration- Study Notes- New syllabus
Edexcel iGCSE Chemistry -2.8C Reactivity Trend and Electronic Configuration- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
2.8C explain the trend in reactivity in Group 7 in terms of electronic configurations
2.8C Explaining the Trend in Reactivity in Group 7 Using Electronic Configurations
Group 7 elements are called the halogens. They include fluorine, chlorine, bromine and iodine.
Reactivity decreases down Group 7. This trend can be explained using electronic configurations and atomic structure.
Electronic Structure of Halogens
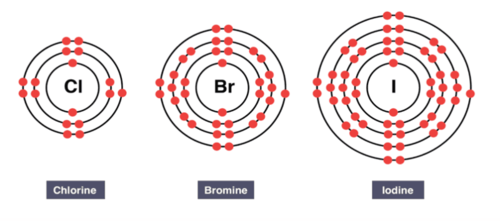
- Fluorine: \( 2,7 \)
- Chlorine: \( 2,8,7 \)
- Bromine: \( 2,8,18,7 \)
- Iodine: \( 2,8,18,18,7 \)
All halogens have seven electrons in their outer shell.
They react by gaining one electron to form a negative ion:
\( \mathrm{X + e^- \rightarrow X^-} \)
Why Halogens React
- Halogens need one electron to achieve a full outer shell.
- They gain one electron in chemical reactions.
- The ease of gaining this electron determines their reactivity.
Explanation of the Trend Down the Group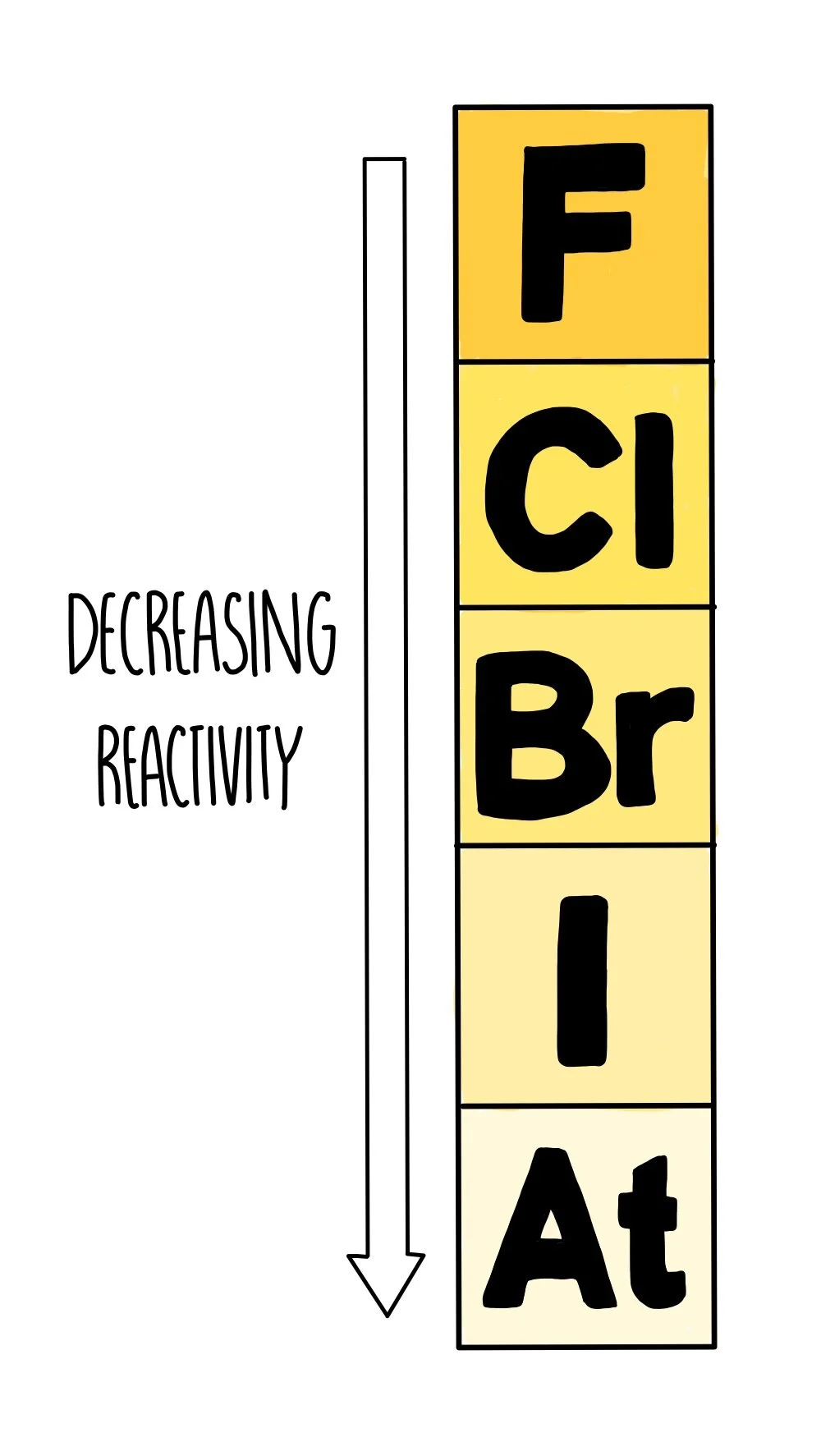
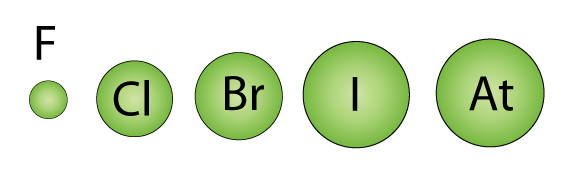
- Down the group, the number of electron shells increases.
- Atomic radius increases.
- Inner electron shells cause increased shielding.
- The attraction between the nucleus and an incoming electron decreases.
- It becomes harder to gain an electron.
- Therefore reactivity decreases down the group.
| Element | Electronic Configuration | Shielding | Attraction for Electron | Reactivity |
|---|---|---|---|---|
| Fluorine | \( 2,7 \) | Least | Strongest | Highest |
| Chlorine | \( 2,8,7 \) | More | Strong | High |
| Bromine | \( 2,8,18,7 \) | Greater | Weaker | Lower |
| Iodine | \( 2,8,18,18,7 \) | Greatest | Weakest | Lowest |
Example 1 (Conceptual):
Why does fluorine have the highest reactivity in Group 7?
▶️ Answer/Explanation
It has the smallest atomic radius.
There is least shielding.
The nucleus strongly attracts an incoming electron.
It gains an electron most easily.
Example 2 (Application):
Which is more reactive, chlorine or bromine? Explain using electronic configuration.
▶️ Answer/Explanation
Chlorine is more reactive.
It has fewer electron shells than bromine.
There is less shielding.
The attraction for an extra electron is stronger.
Example 3 (Hard):
Explain fully why reactivity decreases down Group 7 in terms of electronic configurations and atomic structure.
▶️ Answer/Explanation
All halogens have seven electrons in their outer shell and react by gaining one electron.
Down the group, the number of electron shells increases.
This increases atomic radius and shielding.
The attraction between the nucleus and an incoming electron decreases.
It becomes harder for the atom to gain an electron.
Therefore reactivity decreases down Group 7.
