Edexcel iGCSE Chemistry -3.1 Exothermic and Endothermic Reactions- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -3.1 Exothermic and Endothermic Reactions- Study Notes- New syllabus
Edexcel iGCSE Chemistry -3.1 Exothermic and Endothermic Reactions- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
3.1 know that chemical reactions in which heat energy is given out are described as exothermic, and those in which heat energy is taken in are described as endothermic
3.1 Exothermic and Endothermic Reactions
Definition:
A chemical reaction in which heat energy is given out to the surroundings is called exothermic.
A chemical reaction in which heat energy is taken in from the surroundings is called endothermic.
Energy Transfer in Reactions
During a reaction, bonds are broken and new bonds are formed.
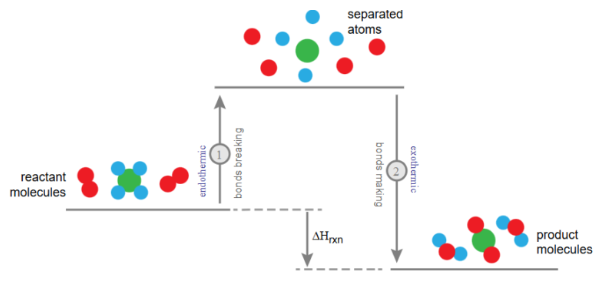
- Energy is required to break bonds.
- Energy is released when new bonds form.
Whether a reaction is exothermic or endothermic depends on which energy change is greater.
Exothermic Reactions
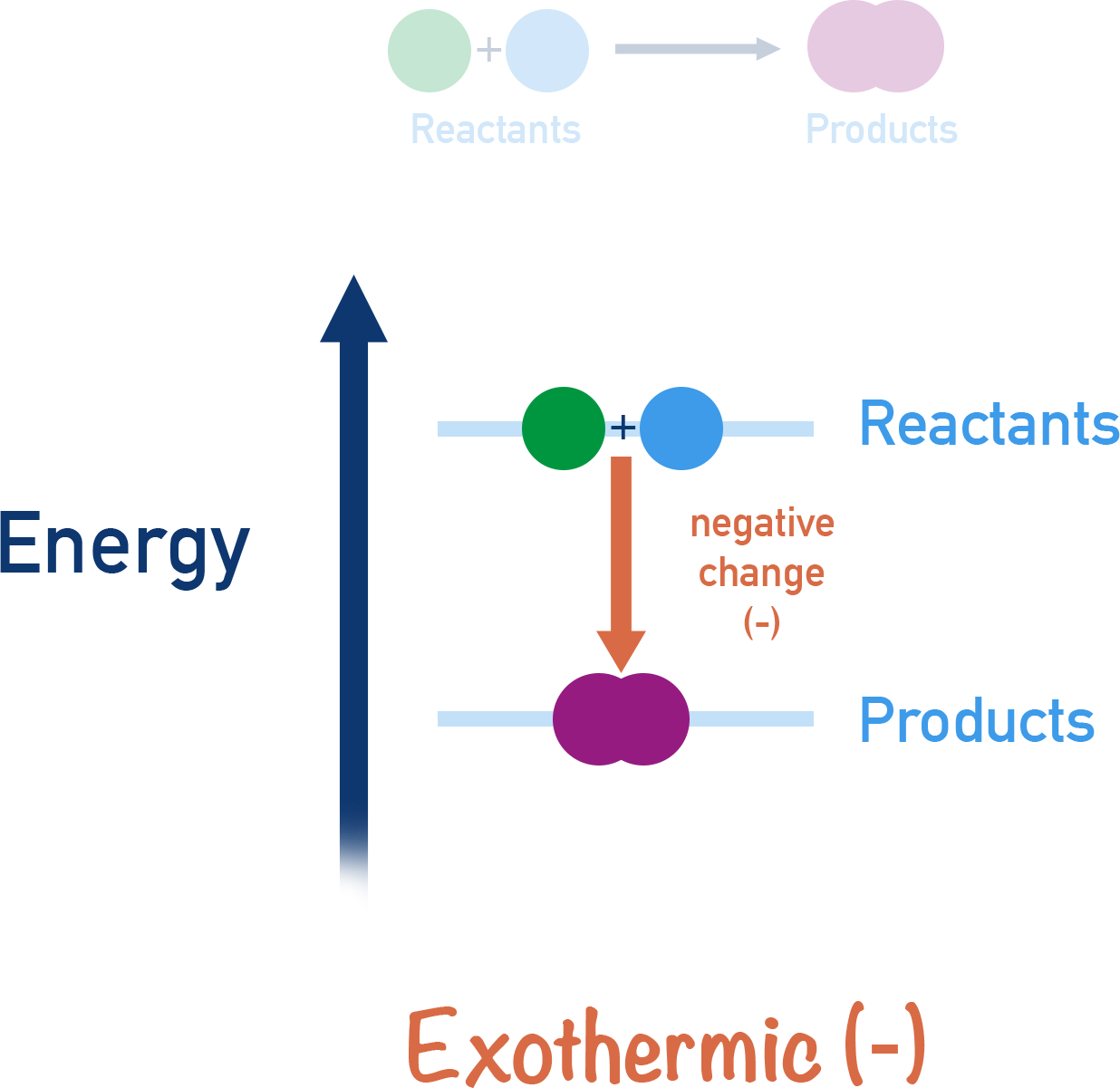
- Release heat to the surroundings.
- Temperature of surroundings increases.
- Products have lower energy than reactants.
Examples:

Combustion of methane:
\( \mathrm{CH_4 + 2O_2 \rightarrow CO_2 + 2H_2O} \)
Neutralisation reactions are also exothermic.
Endothermic Reactions
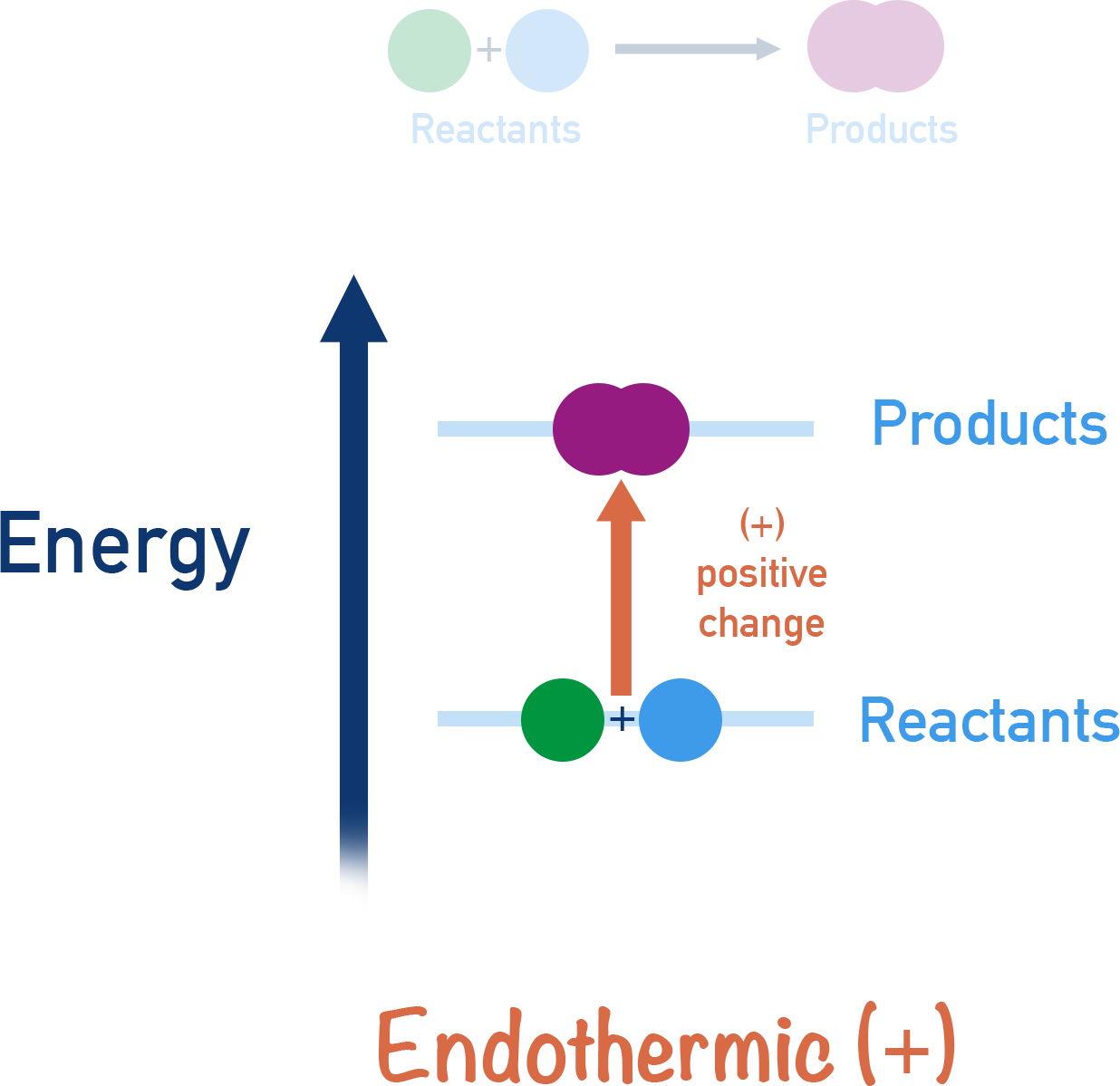
- Absorb heat from the surroundings.
- Temperature of surroundings decreases.
- Products have higher energy than reactants.
Examples:
- Photosynthesis
- Thermal decomposition reactions
\( \mathrm{CaCO_3 \rightarrow CaO + CO_2} \)
| Feature | Exothermic | Endothermic |
|---|---|---|
| Heat flow | Released | Absorbed |
| Temperature change | Increases | Decreases |
| Relative energy of products | Lower | Higher |
Energy Level Diagram Understanding
In exothermic reactions, the energy level of the products is lower than that of the reactants.
In endothermic reactions, the energy level of the products is higher than that of the reactants.
Example 1 (Conceptual):
A reaction causes the temperature of the surroundings to rise. Is it exothermic or endothermic?
▶️ Answer/Explanation
The surroundings gain heat.
The reaction releases heat.
Therefore it is exothermic.
Example 2 (Application):
Classify the thermal decomposition of calcium carbonate:
\( \mathrm{CaCO_3 \rightarrow CaO + CO_2} \)
▶️ Answer/Explanation
Heat must be supplied.
The reaction absorbs heat.
Therefore it is endothermic.
Example 3 (Hard):
Explain fully, in terms of energy transfer and bond breaking/forming, why combustion reactions are exothermic.
▶️ Answer/Explanation
Energy is required to break bonds in the reactants.
New bonds form in the products.
More energy is released when new bonds form than is absorbed breaking bonds.
The excess energy is transferred to the surroundings as heat.
Therefore combustion reactions are exothermic.
