Edexcel iGCSE Chemistry -3.11 Collision Theory- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -3.11 Collision Theory- Study Notes- New syllabus
Edexcel iGCSE Chemistry -3.11 Collision Theory- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
3.11 explain the effects of changes in surface area of a solid, concentration of a solution, pressure of a gas and temperature on the rate of a reaction in terms of particle collision theory
3.11 Explaining Rate Changes Using Collision Theory
Collision Theory:
Chemical reactions occur when particles collide with:
![]()
- Sufficient energy (greater than activation energy).
- The correct orientation.
![]()
Only collisions that meet both conditions are called successful collisions.
The rate of reaction depends on how often successful collisions occur.
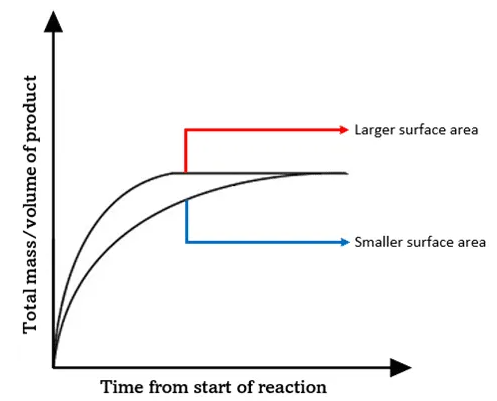
1. Surface Area of a Solid
Effect:
- Increasing surface area increases reaction rate.
Collision Theory Explanation:
- More solid particles are exposed.
- More particles are available for collisions.
- Collision frequency increases.
- Number of successful collisions per second increases.
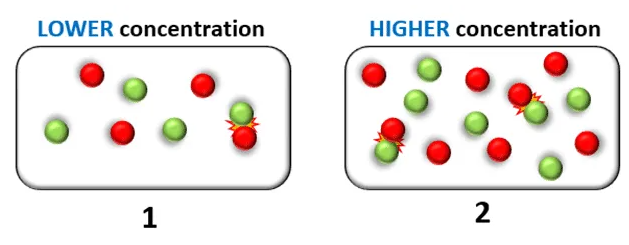
2. Concentration of a Solution
Effect:
- Increasing concentration increases rate.
Collision Theory Explanation:
- More particles per unit volume.
- Particles are closer together.
- Collisions occur more frequently.
- More successful collisions occur per second.
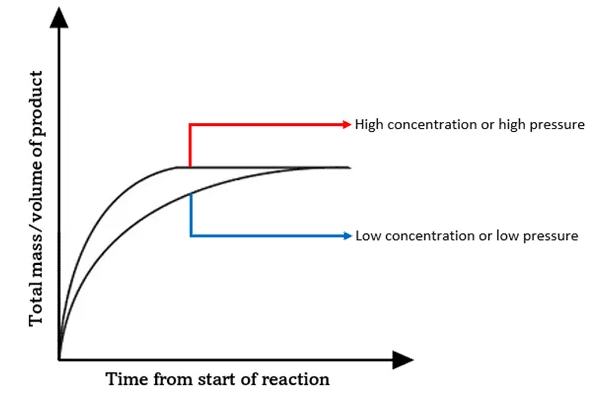
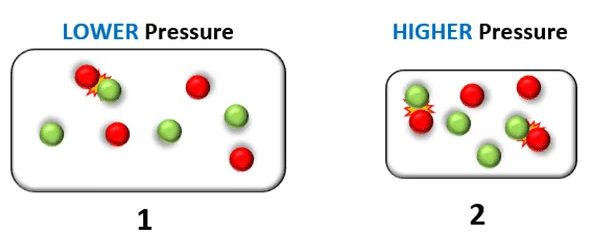
3. Pressure of a Gas
Effect:
- Increasing pressure increases rate (for gases).
Collision Theory Explanation:
- Gas particles are forced closer together.
- Collision frequency increases.
- More successful collisions occur per second.
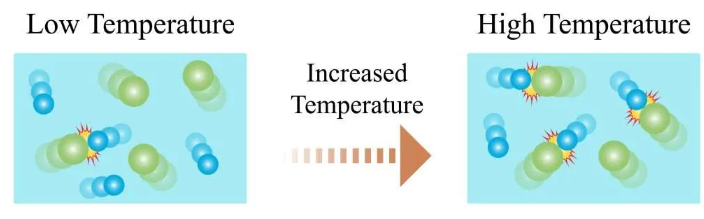
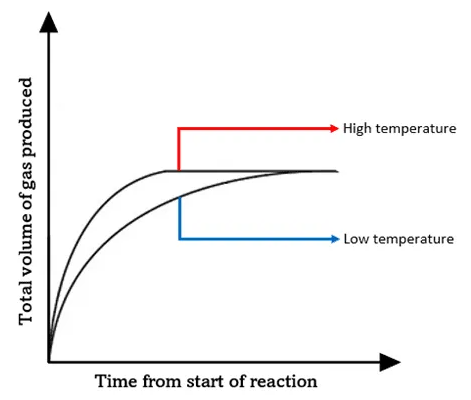
4. Temperature
Effect:
- Increasing temperature increases rate.
Collision Theory Explanation:
- Particles gain kinetic energy.
- They move faster.
- Collisions occur more frequently.
- A greater proportion of particles have energy greater than activation energy.
- More collisions are successful.
| Factor | Collision Frequency | Energy of Particles | Successful Collisions |
|---|---|---|---|
| Surface area ↑ | Increases | No change | Increase |
| Concentration ↑ | Increases | No change | Increase |
| Pressure ↑ (gas) | Increases | No change | Increase |
| Temperature ↑ | Increases | Increases | Increase greatly |
Example 1 (Conceptual):
Why does increasing concentration not change the activation energy?
▶️ Answer/Explanation
Activation energy is a property of the reaction pathway.
Changing concentration only changes collision frequency.
It does not change the energy barrier.
Example 2 (Application):
Explain why increasing surface area increases rate but does not change particle energy.
▶️ Answer/Explanation
More particles are exposed.
Collisions occur more frequently.
However temperature is unchanged, so particle energy stays the same.
Example 3 (Hard):
Explain fully, using collision theory, why increasing temperature has a greater effect on rate than increasing concentration.
▶️ Answer/Explanation
Increasing concentration increases collision frequency only.
Particle energy remains the same.
Increasing temperature increases both collision frequency and kinetic energy.
A much larger proportion of particles exceed activation energy.
Therefore the number of successful collisions increases significantly.
This leads to a greater increase in reaction rate.