Edexcel iGCSE Chemistry -3.13 Catalysts and Activation Energy- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -3.13 Catalysts and Activation Energy- Study Notes- New syllabus
Edexcel iGCSE Chemistry -3.13 Catalysts and Activation Energy- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
3.13 know that a catalyst works by providing an alternative pathway with lower activation energy
3.13 How a Catalyst Works: Alternative Pathway and Lower Activation Energy
Key Statement:
A catalyst works by providing an alternative reaction pathway with a lower activation energy.
![]()
Activation Energy (Ea)
Activation energy is the minimum energy that particles must have for a successful collision.
![]()
- It is the energy barrier of a reaction.
- Without sufficient energy, particles will not react.
What a Catalyst Changes (and What It Does Not)
- Lowers activation energy.
- Increases rate of reaction.
- Does not change the overall enthalpy change \( \Delta H \).
- Does not change the energy of reactants or products.
Energy Level Diagram Explanation
- The reactants and products remain at the same energy level.
- The peak of the energy curve (activation energy) is lower with a catalyst.
- The difference between reactants and products ( \( \Delta H \) ) remains unchanged.
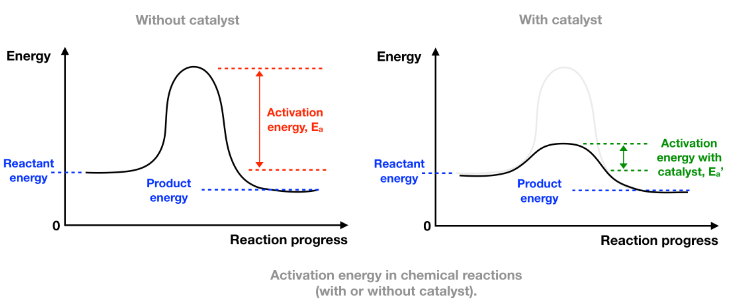
With catalyst: lower \( E_a \)
Without catalyst: higher \( E_a \)
Collision Theory Explanation
- Lower activation energy means the energy barrier is reduced.
- More particles have energy greater than activation energy.
- The number of successful collisions per second increases.
- The reaction happens faster.
| Feature | With Catalyst | Without Catalyst |
|---|---|---|
| Activation energy | Lower | Higher |
| Rate of reaction | Faster | Slower |
| ΔH | Unchanged | Unchanged |
Example 1 (Conceptual):
Why does lowering activation energy increase reaction rate?
▶️ Answer/Explanation
The energy barrier is reduced.
More particles have sufficient energy.
More successful collisions occur per second.
The rate increases.
Example 2 (Application):
Does a catalyst make an endothermic reaction exothermic?
▶️ Answer/Explanation
No.
The overall enthalpy change \( \Delta H \) does not change.
Only the activation energy changes.
Example 3 (Hard):
Explain fully, using an energy diagram and collision theory, how a catalyst increases the rate of reaction.
▶️ Answer/Explanation
The catalyst provides an alternative reaction pathway.
This pathway has lower activation energy.
On the energy diagram, the peak is lower.
The reactant and product energy levels remain the same.
A greater proportion of particles have energy greater than activation energy.
Therefore more successful collisions occur per second.
The catalyst is chemically unchanged at the end.
