Edexcel iGCSE Chemistry -3.15 Practical: Rate of Reaction with Marble Chips- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -3.15 Practical: Rate of Reaction with Marble Chips- Study Notes- New syllabus
Edexcel iGCSE Chemistry -3.15 Practical: Rate of Reaction with Marble Chips- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
3.15 practical: investigate the effect of changing the surface area of marble chips and of changing the concentration of hydrochloric acid on the rate of reaction between marble chips and dilute hydrochloric acid
3.15 Practical: Effect of Surface Area and Concentration on the Rate of Reaction
Aim:
To investigate how changing the surface area of marble chips and the concentration of hydrochloric acid affects the rate of reaction.
Reaction Used
\( \mathrm{CaCO_3(s) + 2HCl(aq) \rightarrow CaCl_2(aq) + CO_2(g) + H_2O(l)} \)
Carbon dioxide gas is produced. The rate can be measured by:
- Volume of \( \mathrm{CO_2} \) produced over time.
- Loss of mass over time.
Part A: Effect of Surface Area
Independent Variable: Size of marble chips (large chips vs powdered marble). 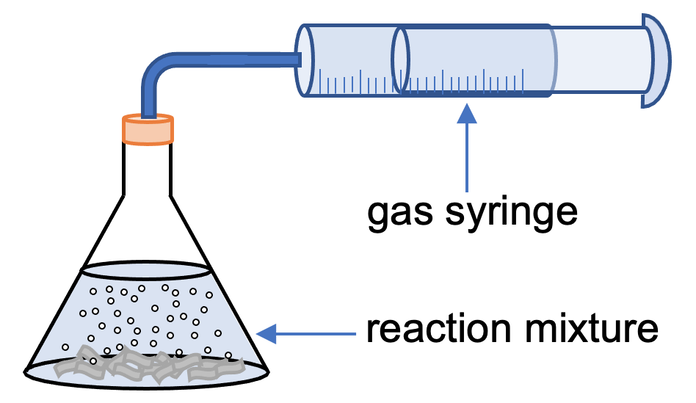
Control Variables:
- Mass of calcium carbonate.
- Volume of acid.
- Concentration of acid.
- Temperature.
Method:
- Measure a fixed volume of dilute hydrochloric acid into a conical flask.
- Add a known mass of marble chips.
- Quickly attach gas syringe.
- Record volume of \( \mathrm{CO_2} \) every 10 seconds.
- Repeat using powdered marble with same mass.
Expected Result: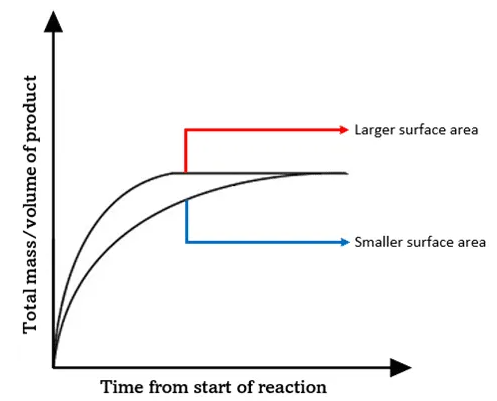
- Powder reacts faster.
- Steeper graph of volume vs time.
Explanation (Collision Theory):
- Greater surface area exposed.
- More frequent collisions between acid particles and solid.
- More successful collisions per second.
Part B: Effect of Acid Concentration
Independent Variable: Concentration of \( \mathrm{HCl} \).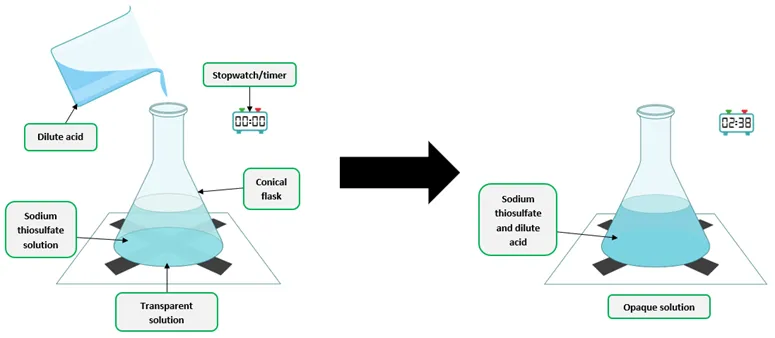
Control Variables:
- Mass of marble chips.
- Size of marble chips.
- Volume of solution.
- Temperature.
Method:
- Prepare different concentrations of hydrochloric acid.
- Add identical mass of marble chips to each.
- Measure volume of gas produced over time.
Expected Result: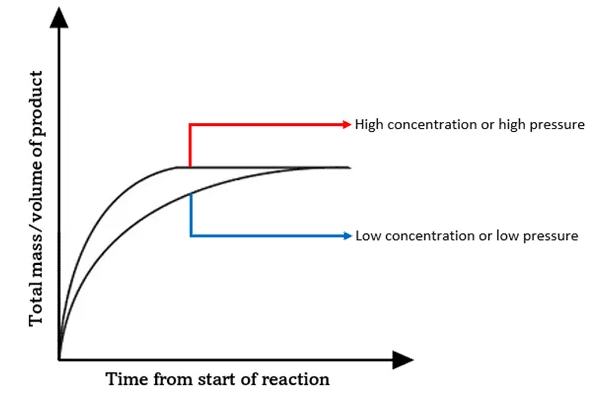
- Higher concentration → faster reaction.
- Steeper initial gradient on graph.
Explanation (Collision Theory):
- More acid particles per unit volume.
- More frequent collisions.
- More successful collisions per second.
Graph Interpretation
- Initial gradient represents rate.
- Steeper gradient → faster reaction.
- Final volume of gas is the same if amount of marble is same.
| Factor Changed | Collision Frequency | Effect on Rate |
|---|---|---|
| Surface area ↑ | Increases | Rate increases |
| Concentration ↑ | Increases | Rate increases |
Sources of Error
- Gas may escape before sealing flask.
- Inconsistent chip sizes.
- Delay in starting stopwatch.
- Temperature changes.
Example 1 (Conceptual):
Why does powdered marble react faster than large chips?
▶️ Answer/Explanation
Powder has larger surface area.
More particles are exposed.
Collisions occur more frequently.
The reaction rate increases.
Example 2 (Application):
If concentration is doubled, what happens to the initial gradient of the graph?
▶️ Answer/Explanation
The gradient becomes steeper.
This shows a faster rate of reaction.
Example 3 (Hard):
Explain fully, using collision theory, why increasing concentration increases the rate of reaction between marble chips and hydrochloric acid.
▶️ Answer/Explanation
Increasing concentration increases the number of acid particles per unit volume.
Particles are closer together.
Collisions between acid particles and the marble surface occur more frequently.
The number of successful collisions per second increases.
Therefore the reaction rate increases.
