Edexcel iGCSE Chemistry -3.16 Practical: Catalytic Decomposition of Hydrogen Peroxide- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -Link- Study Notes- New syllabus
Edexcel iGCSE Chemistry -Link- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
3.16 practical: investigate the effect of different solids on the catalytic decomposition of hydrogen peroxide solution
3.16 Practical: Investigating the Effect of Different Solids on the Catalytic Decomposition of Hydrogen Peroxide
Aim:
To investigate how different solid substances affect the rate of decomposition of hydrogen peroxide.
Reaction
\( \mathrm{2H_2O_2(aq) \rightarrow 2H_2O(l) + O_2(g)} \)
Hydrogen peroxide decomposes slowly on its own, but certain solids act as catalysts and increase the rate.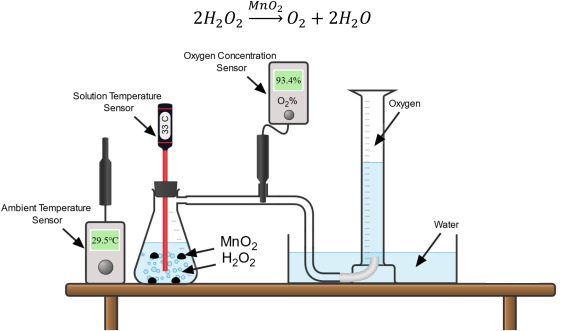
Possible Catalysts to Test
- Manganese dioxide \( \mathrm{MnO_2} \)
- Copper(II) oxide \( \mathrm{CuO} \)
- Iron(III) oxide \( \mathrm{Fe_2O_3} \)
- No solid (control experiment)
Apparatus
- Conical flask
- Gas syringe (or inverted measuring cylinder)
- Hydrogen peroxide solution
- Solid catalysts
- Stopwatch
- Balance
- Rubber bung and delivery tube
- Safety goggles
Variables
- Independent variable: Type of solid used.
- Dependent variable: Volume of oxygen produced per unit time.
- Control variables:
- Volume of hydrogen peroxide.
- Concentration of hydrogen peroxide.
- Mass of solid catalyst.
- Temperature.
Method
- Measure a fixed volume of hydrogen peroxide into a conical flask.
- Add a measured mass of the solid catalyst.
- Immediately fit the bung connected to a gas syringe.
- Start the stopwatch.
- Record volume of oxygen every 10 seconds.
- Repeat using different solids.
Expected Results
- \( \mathrm{MnO_2} \) gives fastest rate.
- Some solids may give slower rates.
- Without catalyst, reaction is very slow.
The best catalyst produces the steepest initial gradient on the graph of volume vs time.
Collision Theory Explanation
- A catalyst provides an alternative reaction pathway.
- This pathway has lower activation energy \( E_a \).
- A greater proportion of particles have sufficient energy.
- More successful collisions occur per second.
The catalyst remains chemically unchanged at the end.
| Solid Used | Relative Rate | Initial Gradient |
|---|---|---|
| None | Very slow | Small |
| \( \mathrm{CuO} \) | Moderate | Medium |
| \( \mathrm{MnO_2} \) | Fast | Steep |
Sources of Error
- Gas may escape before bung is fitted.
- Different particle sizes of solid affect surface area.
- Inconsistent timing.
- Temperature fluctuations.
Example 1 (Conceptual):
Why does manganese dioxide increase the rate of decomposition?
▶️ Answer/Explanation
It provides an alternative pathway.
The activation energy is lowered.
More successful collisions occur.
Example 2 (Application):
Which solid would produce the steepest graph of volume vs time?
▶️ Answer/Explanation
The most effective catalyst.
Typically \( \mathrm{MnO_2} \).
Example 3 (Hard):
Explain fully, using collision theory and activation energy, why adding a catalyst increases the rate of decomposition of hydrogen peroxide.
▶️ Answer/Explanation
The catalyst provides an alternative reaction pathway.
This pathway has lower activation energy \( E_a \).
More particles have energy greater than activation energy.
The number of successful collisions per second increases.
Therefore the reaction rate increases.
The catalyst is chemically unchanged at the end.
