Edexcel iGCSE Chemistry -3.17–3.18 Reversible Reactions and Examples- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -3.17–3.18 Reversible Reactions and Examples- Study Notes- New syllabus
Edexcel iGCSE Chemistry -3.17–3.18 Reversible Reactions and Examples- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
3.17 know that some reactions are reversible and this is indicated by the symbol ⇌ in equations
3.18 describe reversible reactions such as the dehydration of hydrated copper(II) sulfate and the effect of heat on ammonium
chloride
3.17 Reversible Reactions and the Symbol \( \rightleftharpoons \)
Definition:
A reversible reaction is a reaction in which the products can react to reform the original reactants.
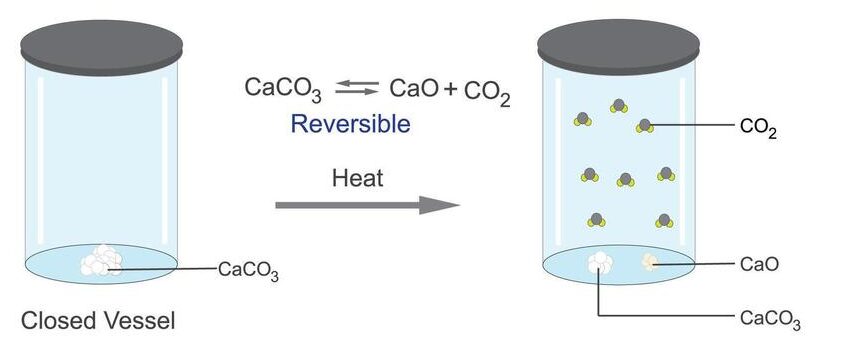
Reversible reactions are shown using the symbol:
\( \rightleftharpoons \)
This symbol shows that the reaction can occur in both directions.
Irreversible vs Reversible
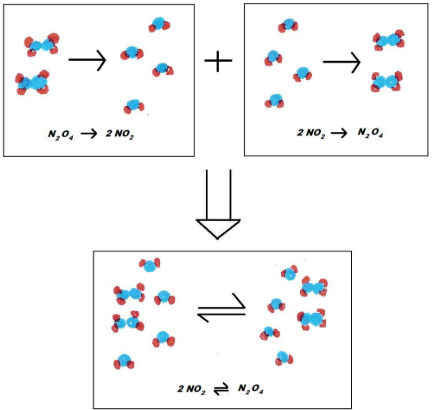
- Irreversible reactions use a single arrow \( \rightarrow \).
- Reversible reactions use the double arrow \( \rightleftharpoons \).
Example of a Reversible Reaction
\( \mathrm{NH_4Cl(s) \rightleftharpoons NH_3(g) + HCl(g)} \)
When heated, ammonium chloride decomposes. When cooled, the gases react to form solid ammonium chloride again.
Dynamic Equilibrium (Foundation Understanding)
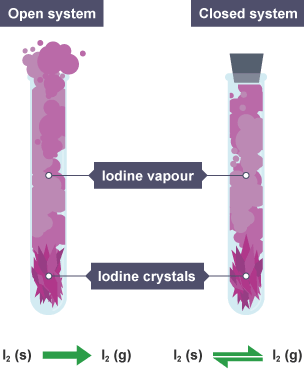
In a closed system:
- The forward reaction and reverse reaction occur simultaneously.
- At equilibrium, both occur at the same rate.
- The concentrations of reactants and products remain constant.
| Type of Reaction | Symbol Used | Can Go Backwards? |
|---|---|---|
| Irreversible | \( \rightarrow \) | No |
| Reversible | \( \rightleftharpoons \) | Yes |
Example 1 (Conceptual):
What does the symbol \( \rightleftharpoons \) show in a chemical equation?
▶️ Answer/Explanation
The reaction is reversible.
It can occur in both directions.
Products can reform reactants.
Example 2 (Application):
Write an equation showing that the reaction between ammonia and hydrogen chloride is reversible.
▶️ Answer/Explanation
\( \mathrm{NH_3(g) + HCl(g) \rightleftharpoons NH_4Cl(s)} \)
The double arrow shows it is reversible.
Example 3 (Hard):
Explain fully what happens in a reversible reaction at dynamic equilibrium.
▶️ Answer/Explanation
The forward and reverse reactions both occur.
They occur at the same rate.
The concentrations of reactants and products remain constant.
The system must be closed.
The reaction continues, but there is no overall change.
3.18 Examples of Reversible Reactions
Some reactions are reversible, meaning the products can react to reform the original reactants.
Reversible reactions are shown using: \( \rightleftharpoons \)
1. Dehydration of Hydrated Copper(II) Sulfate
Reaction:
\( \mathrm{CuSO_4 \cdot 5H_2O(s) \rightleftharpoons CuSO_4(s) + 5H_2O(g)} \)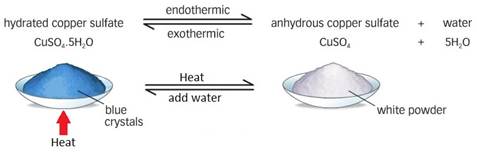
Forward Reaction (Heating):
- Blue hydrated copper(II) sulfate is heated.
- Water of crystallisation is removed.
- White anhydrous copper(II) sulfate is formed.
Reverse Reaction (Adding Water):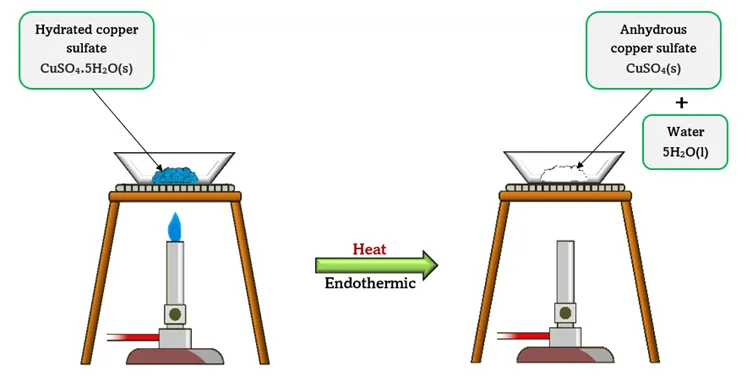
- Water is added to white anhydrous copper(II) sulfate.
- The blue colour returns.
- The hydrated compound reforms.
Key Idea:
- The reaction is reversible.
- Heat removes water.
- Adding water reverses the reaction.
2. Effect of Heat on Ammonium Chloride
Reaction:
\( \mathrm{NH_4Cl(s) \rightleftharpoons NH_3(g) + HCl(g)} \)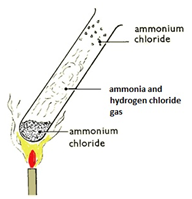
Forward Reaction (Heating):
- Solid ammonium chloride is heated.
- It decomposes into ammonia gas and hydrogen chloride gas.
- White fumes may be observed.
Reverse Reaction (Cooling):
- When gases cool, they recombine.
- Solid ammonium chloride reforms.
This often appears as white solid forming on cooler parts of the apparatus.
| Reaction | Effect of Heat | Reverse Reaction |
|---|---|---|
| Hydrated \( \mathrm{CuSO_4} \) | Turns blue to white | Add water → blue returns |
| \( \mathrm{NH_4Cl} \) | Forms \( \mathrm{NH_3} \) and \( \mathrm{HCl} \) gases | Cooling reforms solid |
Example 1 (Conceptual):
Why does anhydrous copper(II) sulfate turn blue when water is added?
▶️ Answer/Explanation
The reverse reaction occurs.
Water molecules are added back.
Hydrated copper(II) sulfate reforms.
The blue colour returns.
Example 2 (Application):
What happens when ammonium chloride is heated strongly?
▶️ Answer/Explanation
It decomposes.
\( \mathrm{NH_4Cl(s) \rightarrow NH_3(g) + HCl(g)} \)
The reaction is reversible.
Example 3 (Hard):
Explain fully why the dehydration of hydrated copper(II) sulfate is described as reversible.
▶️ Answer/Explanation
When heated, water of crystallisation is removed.
\( \mathrm{CuSO_4 \cdot 5H_2O \rightarrow CuSO_4 + 5H_2O} \)
The blue solid becomes white.
When water is added, the reverse reaction occurs.
The hydrated compound reforms.
Because the reaction can go in both directions, it is reversible.
