Edexcel iGCSE Chemistry -3.19–3.20C Dynamic Equilibrium and Its Characteristics- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -3.19–3.20C Dynamic Equilibrium and Its Characteristics- Study Notes- New syllabus
Edexcel iGCSE Chemistry -3.19–3.20C Dynamic Equilibrium and Its Characteristics- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
3.19C know that a reversible reaction can reach dynamic equilibrium in a sealed container
3.20C know that the characteristics of a reaction at dynamic equilibrium are:
• the forward and reverse reactions occur at the same rate
• the concentrations of reactants and products remain constant
3.19C Dynamic Equilibrium in a Sealed Container
A reversible reaction can reach dynamic equilibrium in a sealed container.
What is Dynamic Equilibrium?
Dynamic equilibrium occurs when:

- The forward reaction and reverse reaction occur at the same rate.
- The concentrations of reactants and products remain constant.
- The reaction is taking place in a closed (sealed) system.
Even though concentrations remain constant, reactions are still occurring. This is why it is called dynamic.
Why a Sealed Container is Needed
![]()
- No substances can enter or leave the system.
- Reactants are not lost.
- Products cannot escape.
- This allows the reverse reaction to occur.
If gases escape, equilibrium cannot be established.
Example
\( \mathrm{NH_3(g) + HCl(g) \rightleftharpoons NH_4Cl(s)} \)
In a sealed container:
- Ammonia and hydrogen chloride react to form ammonium chloride.
- Some ammonium chloride decomposes back into gases.
- Eventually, the forward and reverse reactions occur at equal rates.
Important Features of Dynamic Equilibrium
- Occurs only in a closed system.
- No overall change in concentration.
- Both reactions continue.
- Rates of forward and reverse reactions are equal.
| Condition | At Dynamic Equilibrium |
|---|---|
| System type | Sealed (closed) |
| Forward rate | Equal to reverse rate |
| Concentrations | Constant |
| Reaction activity | Still occurring |
Example 1 (Conceptual):
Why must the container be sealed for equilibrium to be established?
▶️ Answer/Explanation
If products escape, the reverse reaction cannot occur properly.
A closed system is needed.
This allows rates to become equal.
Example 2 (Application):
At equilibrium, are the amounts of reactants and products equal?
▶️ Answer/Explanation
No.
The rates are equal, not necessarily the amounts.
Concentrations remain constant.
Example 3 (Hard):
Explain fully what is meant by dynamic equilibrium in a reversible reaction.
▶️ Answer/Explanation
Dynamic equilibrium occurs in a closed system.
The forward and reverse reactions continue.
They occur at the same rate.
There is no overall change in concentration.
The reaction is dynamic because particles are still reacting.
3.20C Characteristics of Dynamic Equilibrium
Definition:
Dynamic equilibrium is the state reached in a reversible reaction (in a sealed container) where the forward and reverse reactions occur at the same rate.
Two Key Characteristics
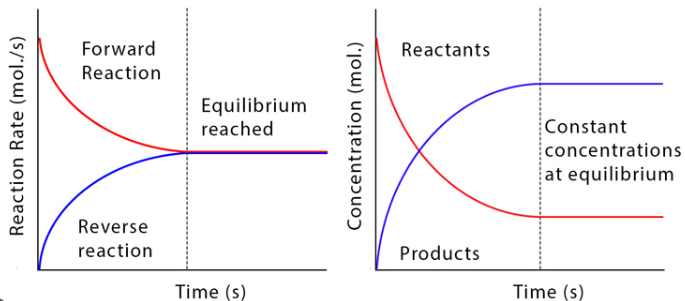
- The forward and reverse reactions occur at the same rate.
- The concentrations of reactants and products remain constant.
It is important to understand that constant concentration does not mean the reaction has stopped.
1. Equal Rates
At equilibrium:
Rate of forward reaction = Rate of reverse reaction
- Reactants are forming products.
- Products are reforming reactants.
- Both processes happen simultaneously.
2. Constant Concentrations
- The amounts of reactants and products stay the same.
- They are not necessarily equal to each other.
- There is no overall change in measurable quantities.
Example Reaction
\( \mathrm{N_2(g) + 3H_2(g) \rightleftharpoons 2NH_3(g)} \)
At dynamic equilibrium:
- Ammonia is being formed.
- Ammonia is decomposing.
- Both occur at the same rate.
- Concentrations remain constant.
| Feature | At Dynamic Equilibrium |
|---|---|
| System type | Closed / sealed |
| Forward reaction | Occurs |
| Reverse reaction | Occurs |
| Reaction rates | Equal |
| Concentrations | Constant |
Example 1 (Conceptual):
Does dynamic equilibrium mean the reaction has stopped?
▶️ Answer/Explanation
No.
Both forward and reverse reactions are still occurring.
They occur at the same rate.
Example 2 (Application):
At equilibrium, why does the concentration of products not increase further?
▶️ Answer/Explanation
Products are formed at the same rate they are converted back into reactants.
There is no overall change in concentration.
Example 3 (Hard):
Explain fully the two characteristics of a reaction at dynamic equilibrium.
▶️ Answer/Explanation
At dynamic equilibrium, the forward and reverse reactions occur at the same rate.
This means the speed of product formation equals the speed of product decomposition.
As a result, the concentrations of reactants and products remain constant.
The reaction is dynamic because particles continue to react.
The system must be closed for equilibrium to be maintained.
