Edexcel iGCSE Chemistry -3.21 -3.22C Effect of Catalysts, Temperature and Pressure on Equilibrium- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -3.21 -3.22C Effect of Catalysts, Temperature and Pressure on Equilibrium- Study Notes- New syllabus
Edexcel iGCSE Chemistry -3.21 -3.22C Effect of Catalysts, Temperature and Pressure on Equilibrium- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
3.21C understand why a catalyst does not affect the position of equilibrium in a reversible reaction
3.22C know the effect of changing either temperature or pressure on the position of equilibrium in a reversible reaction:
• an increase (or decrease) in temperature shifts the position of equilibrium in the direction of the endothermic (or exothermic) reaction
• an increase (or decrease) in pressure shifts the position of equilibrium in the direction that produces fewer (or more) moles of gas
(References to Le Chatelier’s principle are not required.)
3.21C Why a Catalyst Does Not Affect the Position of Equilibrium
Key Idea:
A catalyst does not change the position of equilibrium in a reversible reaction.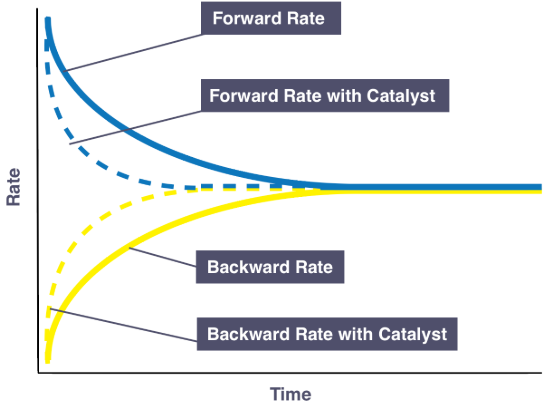
What is the Position of Equilibrium?
The position of equilibrium refers to the relative amounts of reactants and products present at equilibrium.
- If there are more products → equilibrium lies to the right.
- If there are more reactants → equilibrium lies to the left.
What a Catalyst Does

- Provides an alternative pathway.
- Lowers the activation energy \( E_a \).
- Increases the rate of reaction.
- Speeds up both forward and reverse reactions equally.
Why the Position Does Not Change
- The catalyst lowers activation energy for both directions.
- The enthalpy change \( \Delta H \) remains unchanged.
- The energy levels of reactants and products remain unchanged.
- Both reactions speed up equally.
- Therefore, the ratio of reactants to products at equilibrium stays the same.
The catalyst only allows equilibrium to be reached faster.
Energy Profile Explanation

- With a catalyst, activation energy is lower.
- \( \Delta H \) is unchanged.
- The heights of reactants and products stay the same.
| Effect | With Catalyst | Does Position Change? |
|---|---|---|
| Activation energy | Lower | No |
| Forward rate | Faster | No |
| Reverse rate | Faster | No |
| Equilibrium composition | Same | No |
Example 1 (Conceptual):
If a catalyst is added to a system at equilibrium, what happens to the amounts of reactants and products?
▶️ Answer/Explanation
The amounts do not change.
The position of equilibrium remains the same.
The reaction just reaches equilibrium faster.
Example 2 (Application):
Why does a catalyst not change \( \Delta H \) in a reversible reaction?
▶️ Answer/Explanation
\( \Delta H \) depends on the energy difference between reactants and products.
A catalyst does not change these energy levels.
It only lowers activation energy.
Example 3 (Hard):
Explain fully why adding a catalyst does not change the position of equilibrium in a reversible reaction.
▶️ Answer/Explanation
A catalyst provides an alternative pathway with lower activation energy.
It lowers activation energy for both forward and reverse reactions.
Both reactions speed up equally.
The enthalpy change \( \Delta H \) remains unchanged.
The energy levels of reactants and products do not change.
Therefore the equilibrium composition remains the same.
The catalyst only allows equilibrium to be reached more quickly.
3.22C Effect of Temperature and Pressure on the Position of Equilibrium
In a reversible reaction at dynamic equilibrium, changing temperature or pressure can change the position of equilibrium.
The position of equilibrium refers to the relative amounts of reactants and products present at equilibrium.
1. Effect of Temperature
Temperature change affects the position depending on whether the reaction is exothermic or endothermic.
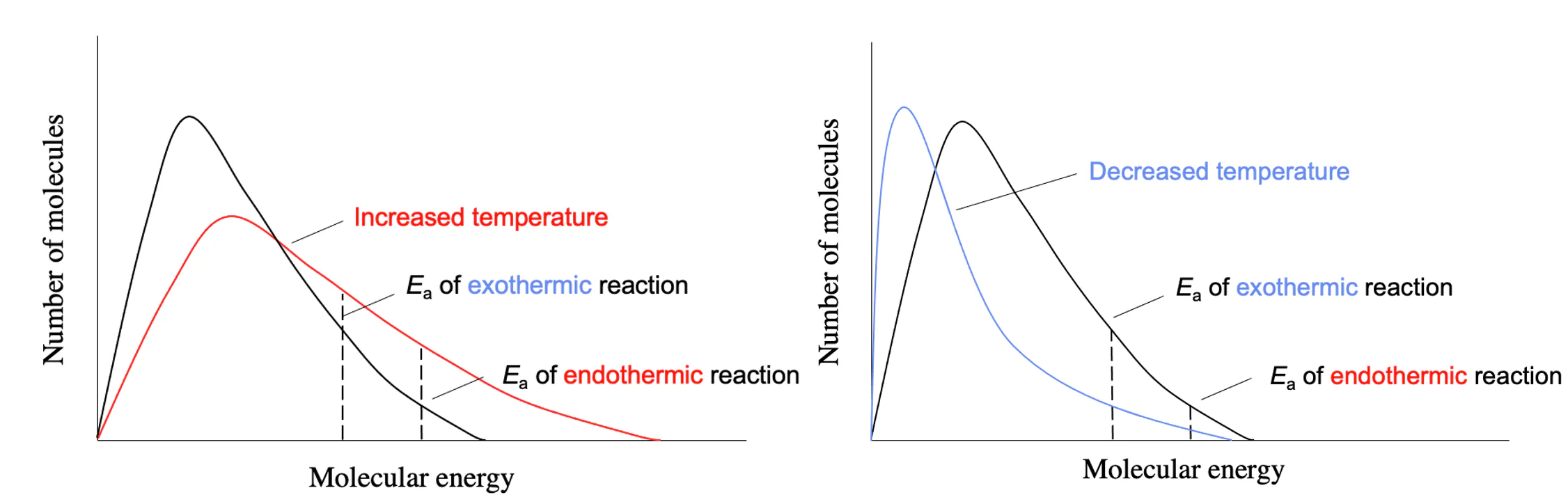
Rules:
- An increase in temperature shifts equilibrium in the direction of the endothermic reaction.
- A decrease in temperature shifts equilibrium in the direction of the exothermic reaction.
This is because increasing temperature supplies heat energy, so the system favours the reaction that absorbs heat.
Example: Haber Process
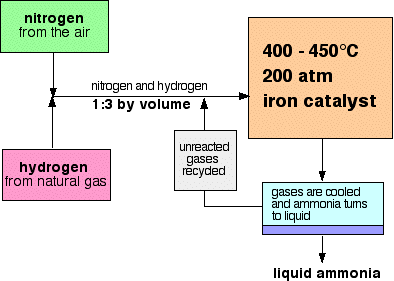
\( \mathrm{N_2(g) + 3H_2(g) \rightleftharpoons 2NH_3(g)} \)
The forward reaction is exothermic.
- Increase in temperature → shifts equilibrium to the left (less \( \mathrm{NH_3} \)).
- Decrease in temperature → shifts equilibrium to the right (more \( \mathrm{NH_3} \)).
2. Effect of Pressure (Gaseous Reactions Only)
Pressure changes only affect equilibria involving gases.
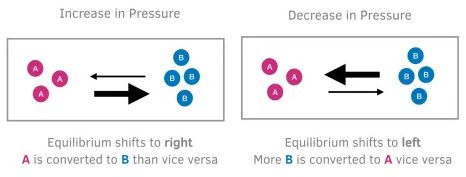
Rules:
- An increase in pressure shifts equilibrium in the direction that produces fewer moles of gas.
- A decrease in pressure shifts equilibrium in the direction that produces more moles of gas.
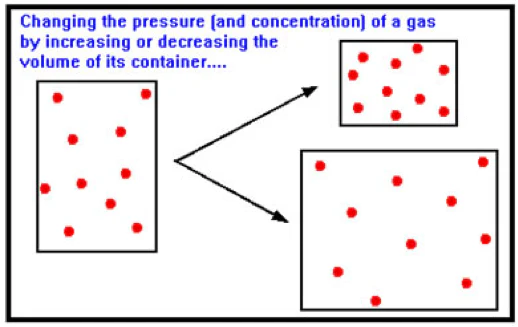
This is because higher pressure favours the side with fewer gas particles.
Haber Process Example (Pressure)
Left side: 4 moles of gas
Right side: 2 moles of gas
- Increase in pressure → shifts to the right (more \( \mathrm{NH_3} \)).
- Decrease in pressure → shifts to the left.
| Change | Shifts Equilibrium Toward |
|---|---|
| Temperature increase | Endothermic direction |
| Temperature decrease | Exothermic direction |
| Pressure increase | Fewer moles of gas |
| Pressure decrease | More moles of gas |
Example 1 (Conceptual):
If a reaction is endothermic in the forward direction, what happens when temperature increases?
▶️ Answer/Explanation
The equilibrium shifts to the right.
The endothermic reaction is favoured.
More products are formed.
Example 2 (Application):
In a gaseous reaction, the left side has 3 moles of gas and the right side has 1 mole. What happens when pressure increases?
▶️ Answer/Explanation
Equilibrium shifts to the right.
This side has fewer moles of gas.
Higher pressure favours fewer gas particles.
Example 3 (Hard):
The forward reaction in an equilibrium is exothermic and produces fewer moles of gas. Explain fully what happens when temperature increases and pressure decreases.
▶️ Answer/Explanation
Increasing temperature favours the endothermic direction.
Since the forward reaction is exothermic, equilibrium shifts to the reverse reaction.
Decreasing pressure favours the side with more moles of gas.
This may also shift equilibrium to the reverse side if it has more moles.
Both changes can reduce the amount of product formed.
