Edexcel iGCSE Chemistry -3.3 Heat Energy Calculations (Q = mcΔT)- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -3.3 Heat Energy Calculations (Q = mcΔT)- Study Notes- New syllabus
Edexcel iGCSE Chemistry -3.3 Heat Energy Calculations (Q = mcΔT)- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
3.3 calculate the heat energy change from a measured temperature change using the expression
Q = mcΔT
3.3 Calculating Heat Energy Change Using \( Q = mc\Delta T \)
Key Formula: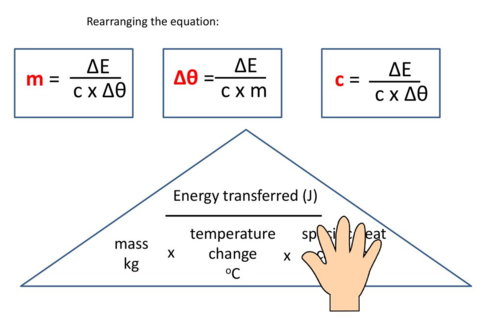
\( Q = mc\Delta T \)
Where:
- \( Q \) = heat energy transferred (J)
- \( m \) = mass of solution (g)
- \( c \) = specific heat capacity \( (4.2 \, \mathrm{J\,g^{-1}\,°C^{-1}}) \)
- \( \Delta T \) = temperature change \( (^\circ\mathrm{C}) \)
Understanding Each Term
- \( m \) is usually the mass of the solution (often assumed equal to volume in cm³).
- \( \Delta T = \text{final temperature} – \text{initial temperature} \).
- If temperature increases → reaction is exothermic.
- If temperature decreases → reaction is endothermic.
Units Reminder
- Mass must be in grams.
- Energy will be in joules.
- Sometimes convert to kJ: \( 1\,\mathrm{kJ} = 1000\,\mathrm{J} \).
Worked Structure (Exam Method)
- Write the formula.
- Substitute values clearly.
- Show units.
- State whether reaction is exothermic or endothermic.
Example 1 (Numerical):
100 g of solution increases in temperature by \( 8^\circ\mathrm{C} \). Calculate the heat energy change.
▶️ Answer/Explanation
Formula:
\( Q = mc\Delta T \)
Substitute values:
\( Q = 100 \times 4.2 \times 8 \)
\( Q = 3360 \, \mathrm{J} \)
Temperature increased → exothermic reaction.
Example 2 (Conceptual):
During a reaction the temperature drops by \( 5^\circ\mathrm{C} \). Is the reaction exothermic or endothermic?
▶️ Answer/Explanation
The temperature decreases.
Heat is absorbed from the surroundings.
The reaction is endothermic.
Example 3 (Hard):
50 cm³ of acid is mixed with 50 cm³ of alkali. The temperature increases from \( 21^\circ\mathrm{C} \) to \( 29^\circ\mathrm{C} \). Calculate the heat energy released.
▶️ Answer/Explanation
Step 1: Calculate total mass.
Total volume = 100 cm³ Assume density = \( 1\,\mathrm{g\,cm^{-3}} \) Mass = 100 g
Step 2: Find \( \Delta T \).
\( \Delta T = 29 – 21 = 8^\circ\mathrm{C} \)
Step 3: Use formula.
\( Q = mc\Delta T \)
\( Q = 100 \times 4.2 \times 8 \)
\( Q = 3360 \, \mathrm{J} \)
The temperature increased, so the reaction is exothermic.
