Edexcel iGCSE Chemistry -3.4 Molar Enthalpy Change- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -3.4 Molar Enthalpy Change- Study Notes- New syllabus
Edexcel iGCSE Chemistry -3.4 Molar Enthalpy Change- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
3.4 calculate the molar enthalpy change (ΔH) from the heat energy change, Q
3.4 Calculating Molar Enthalpy Change \( \Delta H \) from Heat Energy \( Q \)
Definition:
The molar enthalpy change, \( \Delta H \), is the heat energy change when one mole of a substance reacts.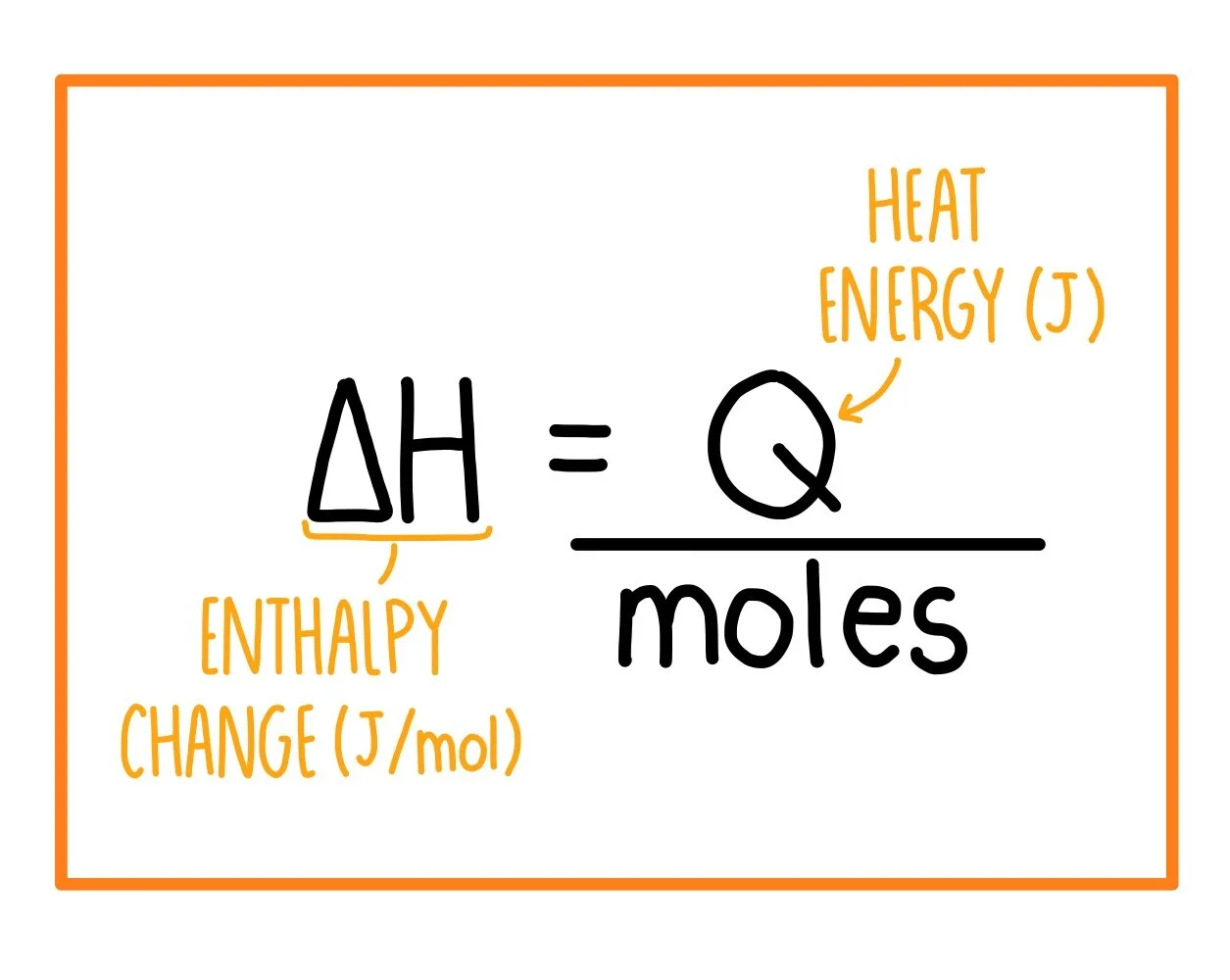
It is usually expressed in:
\( \mathrm{kJ\,mol^{-1}} \)
Key Formula
\( \Delta H = \dfrac{Q}{n} \)
- \( Q \) = heat energy change (in kJ)
- \( n \) = number of moles reacting
Important: If \( Q \) is in joules, convert to kJ first.
Step-by-Step Method
- Calculate \( Q \) using \( Q = mc\Delta T \).
- Convert \( Q \) from J to kJ (divide by 1000).
- Calculate number of moles \( n \).
- Use \( \Delta H = \dfrac{Q}{n} \).
- Add correct sign (+ or −).
- Exothermic → \( \Delta H \) is negative.
- Endothermic → \( \Delta H \) is positive.
Example 1 (Numerical):
50 g of solution increases by \( 6^\circ\mathrm{C} \). 0.50 mol of reactant was used. Calculate \( \Delta H \).
▶️ Answer/Explanation
Step 1: Calculate \( Q \).
\( Q = mc\Delta T \)
\( Q = 50 \times 4.2 \times 6 \)
\( Q = 1260 \, \mathrm{J} \)
Convert to kJ:
\( Q = 1.26 \, \mathrm{kJ} \)
Step 2: Use formula.
\( \Delta H = \dfrac{1.26}{0.50} \)
\( \Delta H = 2.52 \, \mathrm{kJ\,mol^{-1}} \)
Temperature increased → exothermic →
\( \Delta H = -2.52 \, \mathrm{kJ\,mol^{-1}} \)
Example 2 (Conceptual):
Why must heat energy be divided by moles to calculate molar enthalpy change?
▶️ Answer/Explanation
Enthalpy change is defined per mole.
Total heat energy depends on how much reacted.
Dividing by moles gives energy change per mole.
Example 3 (Hard):
100 cm³ of 1.0 mol/dm³ HCl reacts with excess NaOH. Temperature rises from \( 20^\circ\mathrm{C} \) to \( 27^\circ\mathrm{C} \). Calculate the molar enthalpy change of neutralisation.
▶️ Answer/Explanation
Step 1: Find mass of solution.
Volume = 100 cm³ Mass ≈ 100 g
Step 2: Calculate \( \Delta T \).
\( \Delta T = 27 – 20 = 7^\circ\mathrm{C} \)
Step 3: Calculate \( Q \).
\( Q = 100 \times 4.2 \times 7 \)
\( Q = 2940 \, \mathrm{J} = 2.94 \, \mathrm{kJ} \)
Step 4: Calculate moles of HCl.
\( n = cV \)
Volume = 0.100 dm³
\( n = 1.0 \times 0.100 = 0.10 \, \mathrm{mol} \)
Step 5: Calculate \( \Delta H \).
\( \Delta H = \dfrac{2.94}{0.10} \)
\( \Delta H = 29.4 \, \mathrm{kJ\,mol^{-1}} \)
Reaction is exothermic:
\( \Delta H = -29.4 \, \mathrm{kJ\,mol^{-1}} \)
