Edexcel iGCSE Chemistry -3.5C Energy Level Diagrams- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -3.5C Energy Level Diagrams- Study Notes- New syllabus
Edexcel iGCSE Chemistry -3.5C Energy Level Diagrams- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
3.5C draw and explain energy level diagrams to represent exothermic and endothermic reactions
3.5C Energy Level Diagrams for Exothermic and Endothermic Reactions
Purpose of an Energy Level Diagram:
To show the relative energy of reactants and products and the overall enthalpy change \( \Delta H \).
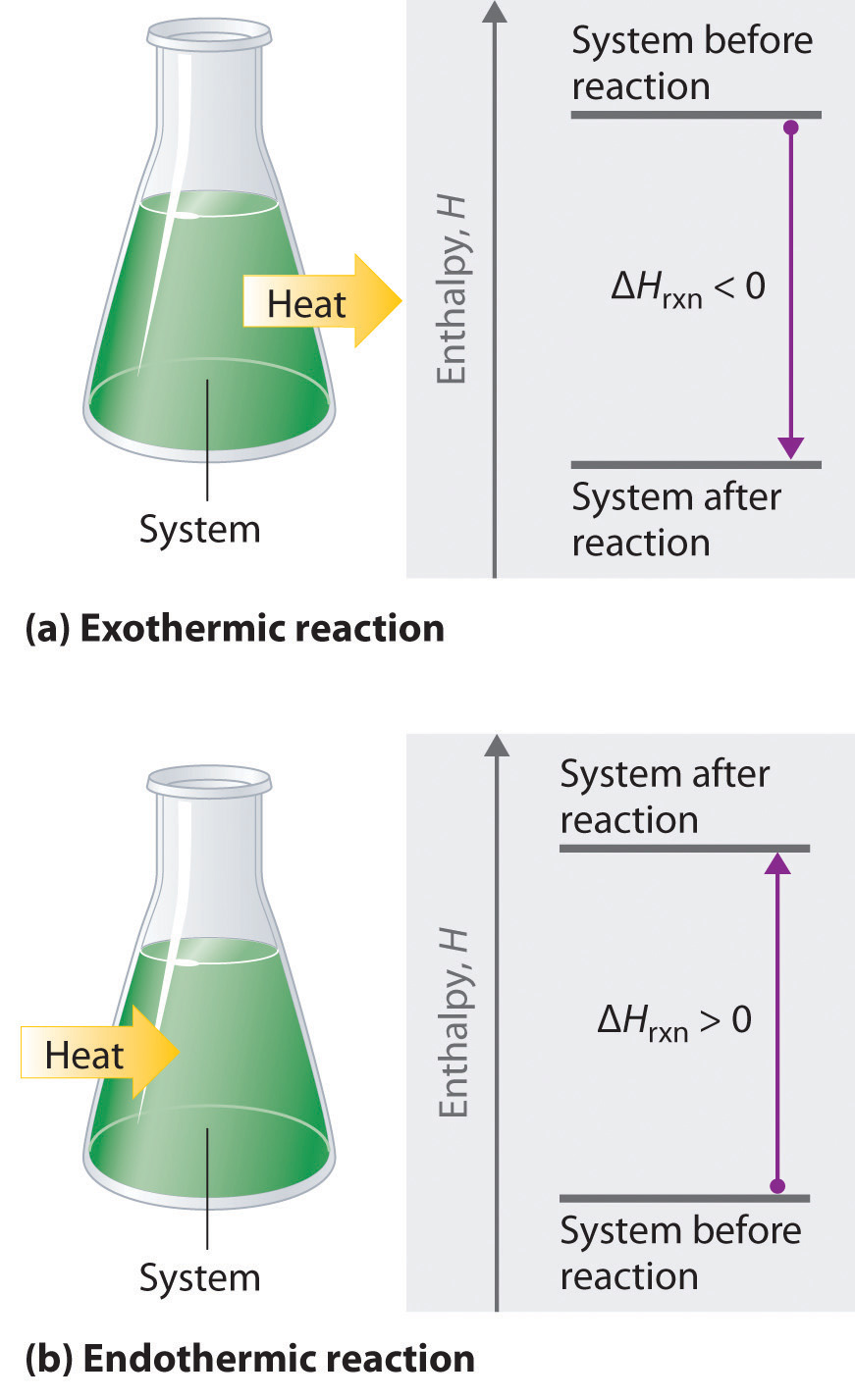
The vertical axis represents energy. The horizontal axis represents progress of reaction.
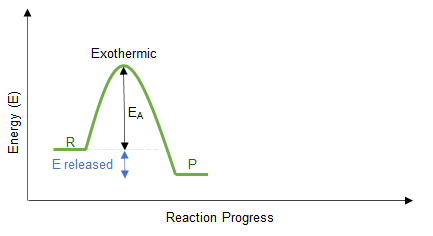
1. Exothermic Reactions
- Energy is released to the surroundings.
- Temperature increases.
- Products have lower energy than reactants.
- \( \Delta H \) is negative.
How to Draw:
- Draw reactants at a higher energy level.
- Draw products at a lower energy level.
- Draw a downward arrow labelled \( \Delta H \).
\( \Delta H < 0 \)
Explanation:
More energy is released when new bonds form than is absorbed breaking bonds. The excess energy is transferred to the surroundings.
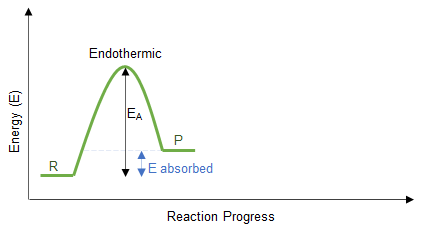
2. Endothermic Reactions
- Energy is absorbed from the surroundings.
- Temperature decreases.
- Products have higher energy than reactants.
- \( \Delta H \) is positive.
How to Draw:
- Draw reactants at a lower energy level.
- Draw products at a higher energy level.
- Draw an upward arrow labelled \( \Delta H \).
\( \Delta H > 0 \)
Explanation:
More energy is absorbed breaking bonds than is released forming new bonds. Energy is taken in from the surroundings.
| Feature | Exothermic | Endothermic |
|---|---|---|
| Energy of Products | Lower | Higher |
| Sign of \( \Delta H \) | Negative | Positive |
| Heat Transfer | Released | Absorbed |
Activation Energy (Extension Understanding)
Both types of reactions require activation energy to start.
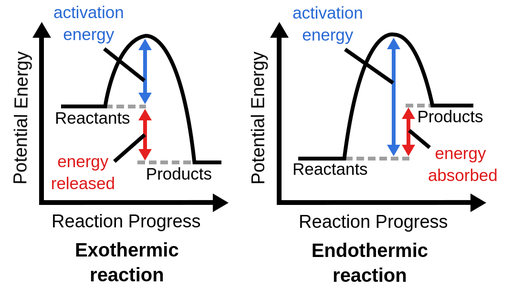
Activation energy is the minimum energy required to start a reaction.
Example 1 (Conceptual):
On an energy diagram, the products are lower than the reactants. What type of reaction is this?
▶️ Answer/Explanation
Products have lower energy.
Energy has been released.
It is exothermic.
\( \Delta H \) is negative.
Example 2 (Drawing):
Explain why \( \Delta H \) is positive in an endothermic reaction.
▶️ Answer/Explanation
The products are at a higher energy level than the reactants.
Energy has been absorbed from the surroundings.
Therefore \( \Delta H \) is positive.
Example 3 (Hard):
Explain fully, using an energy level diagram, why combustion of methane is exothermic.
▶️ Answer/Explanation
The reactants are at a higher energy level.
The products are at a lower energy level.
Energy is released when new bonds form in \( \mathrm{CO_2} \) and \( \mathrm{H_2O} \).
More energy is released than absorbed breaking bonds.
The downward energy difference represents \( \Delta H \).
\( \Delta H \) is negative.