Edexcel iGCSE Chemistry -3.6C Bond Breaking and Bond Making- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -3.6C Bond Breaking and Bond Making- Study Notes- New syllabus
Edexcel iGCSE Chemistry -3.6C Bond Breaking and Bond Making- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
3.6C know that bond-breaking is an endothermic process and that bond-making is an exothermic process
3.6C Bond-Breaking and Bond-Making
Key Principles:
![]()
- Bond-breaking is an endothermic process.
- Bond-making is an exothermic process.
Why Bond-Breaking is Endothermic
Chemical bonds are strong electrostatic attractions between atoms.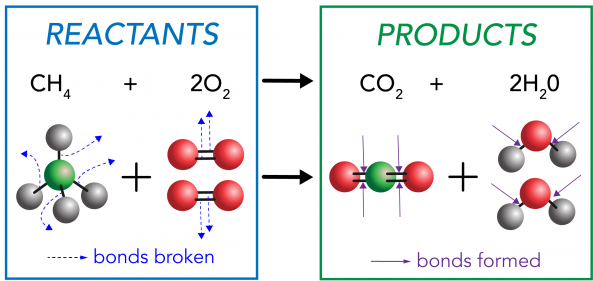
Energy must be supplied to overcome these attractions.
- Energy is absorbed from the surroundings.
- The process requires an input of energy.
- Therefore, bond-breaking is endothermic.
Why Bond-Making is Exothermic
When new bonds form, attractive forces are established between atoms.
- Energy is released to the surroundings.
- The system becomes more stable.
- Therefore, bond-making is exothermic.
Overall Energy Change in Reactions
Every chemical reaction involves:
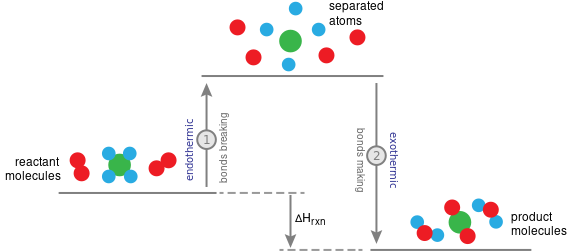
- Breaking bonds in the reactants.
- Making new bonds in the products.
The overall enthalpy change \( \Delta H \) depends on which process involves more energy.
- If energy released making bonds > energy absorbed breaking bonds → exothermic.
- If energy absorbed breaking bonds > energy released making bonds → endothermic.
| Process | Energy Change | Type |
|---|---|---|
| Bond-breaking | Energy absorbed | Endothermic |
| Bond-making | Energy released | Exothermic |
Example: Combustion of Methane
\( \mathrm{CH_4 + 2O_2 \rightarrow CO_2 + 2H_2O} \)
- Bonds in \( \mathrm{CH_4} \) and \( \mathrm{O_2} \) are broken (endothermic).
- New bonds in \( \mathrm{CO_2} \) and \( \mathrm{H_2O} \) are formed (exothermic).
- More energy is released than absorbed.
- Overall reaction is exothermic.
Example 1 (Conceptual):
Why must energy be supplied to break a chemical bond?
▶️ Answer/Explanation
Atoms are held together by strong electrostatic forces.
Energy is needed to overcome these attractions.
Therefore bond-breaking absorbs energy.
Example 2 (Application):
When a bond forms, does energy enter or leave the system?
▶️ Answer/Explanation
Energy is released to the surroundings.
Bond-making is exothermic.
Example 3 (Hard):
Explain fully why some reactions are exothermic even though bond-breaking always requires energy.
▶️ Answer/Explanation
All reactions require energy to break bonds.
This part is endothermic.
However, forming new bonds releases energy.
If more energy is released forming bonds than absorbed breaking bonds,
the overall reaction releases energy.
Therefore the reaction is exothermic.
