Edexcel iGCSE Chemistry -3.7C Bond Energies and Enthalpy Change- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -3.7C Bond Energies and Enthalpy Change- Study Notes- New syllabus
Edexcel iGCSE Chemistry -3.7C Bond Energies and Enthalpy Change- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
3.7C use bond energies to calculate the enthalpy change during a chemical reaction
3.7C Calculating Enthalpy Change Using Bond Energies
Definition:
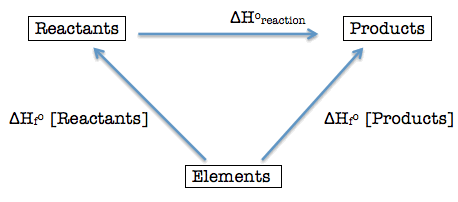
Bond energy is the energy required to break one mole of a particular bond in gaseous molecules.
It is measured in:
\( \mathrm{kJ\,mol^{-1}} \)
Key Formula
\( \Delta H = \text{(Total energy to break bonds)} – \text{(Total energy released forming bonds)} \)
Important:
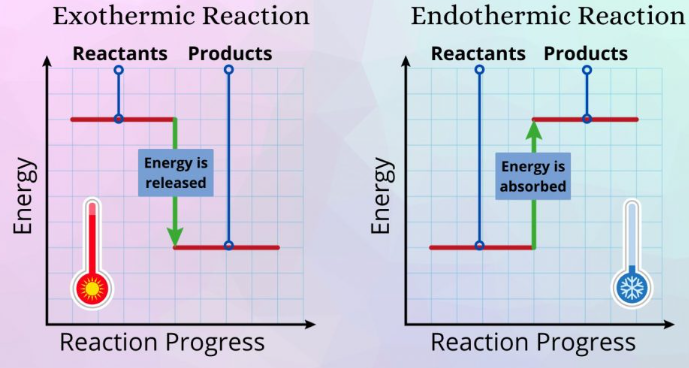
- Bond-breaking is endothermic (energy absorbed).
- Bond-making is exothermic (energy released).
Step-by-Step Method
- Write the balanced equation.
- Identify all bonds broken in the reactants.
- Identify all bonds formed in the products.
- Calculate total energy to break bonds.
- Calculate total energy released forming bonds.
- Use formula \( \Delta H = \text{broken} – \text{formed} \).
- State if reaction is exothermic or endothermic.
Example Bond Energies (Typical Values)
| Bond | Bond Energy (kJ mol⁻¹) |
|---|---|
| \( \mathrm{C-H} \) | 413 |
| \( \mathrm{O=O} \) | 498 |
| \( \mathrm{C=O} \) (in \( \mathrm{CO_2} \)) | 805 |
| \( \mathrm{O-H} \) | 463 |
Example 1 (Numerical):
Calculate \( \Delta H \) for:
\( \mathrm{H_2 + Cl_2 \rightarrow 2HCl} \)
Bond energies:
- \( \mathrm{H-H} = 436 \)
- \( \mathrm{Cl-Cl} = 243 \)
- \( \mathrm{H-Cl} = 431 \)
▶️ Answer/Explanation
Bonds broken:
\( 436 + 243 = 679 \)
Bonds formed:
2 × 431 = 862
Calculate \( \Delta H \):
\( \Delta H = 679 – 862 \)
\( \Delta H = -183 \, \mathrm{kJ\,mol^{-1}} \)
Negative value → exothermic.
Example 2 (Conceptual):
If more energy is needed to break bonds than is released forming bonds, what is the sign of \( \Delta H \)?
▶️ Answer/Explanation
\( \Delta H \) is positive.
The reaction is endothermic.
Example 3 (Hard):
Calculate \( \Delta H \) for:
\( \mathrm{CH_4 + 2O_2 \rightarrow CO_2 + 2H_2O} \)
Using bond energies:
- \( \mathrm{C-H} = 413 \)
- \( \mathrm{O=O} = 498 \)
- \( \mathrm{C=O} = 805 \)
- \( \mathrm{O-H} = 463 \)
▶️ Answer/Explanation
Bonds broken:
4 × 413 (C-H) = 1652
2 × 498 (O=O) = 996
Total broken = 2648
Bonds formed:
2 × 805 (C=O) = 1610
4 × 463 (O-H) = 1852
Total formed = 3462
Calculate:
\( \Delta H = 2648 – 3462 \)
\( \Delta H = -814 \, \mathrm{kJ\,mol^{-1}} \)
Negative value → strongly exothermic.
