Edexcel iGCSE Chemistry -3.8 Practical: Temperature Changes in Reactions- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -3.8 Practical: Temperature Changes in Reactions- Study Notes- New syllabus
Edexcel iGCSE Chemistry -3.8 Practical: Temperature Changes in Reactions- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
3.8 practical: investigate temperature changes accompanying some of the following types of change:
• salts dissolving in water
• neutralisation reactions
• displacement reactions
• combustion reactions
3.8 Practical: Investigating Temperature Changes in Chemical Reactions
Aim:
To measure temperature changes accompanying different types of chemical change and identify whether they are exothermic or endothermic.
Core Principle
- Exothermic → temperature increases
- Endothermic → temperature decreases
Temperature change is measured using a thermometer in an insulated container (usually a polystyrene cup).
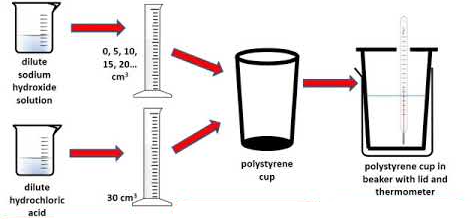
Apparatus (General)
- Polystyrene cup (insulation)
- Beaker
- Thermometer
- Measuring cylinder
- Stirrer
- Balance (if required)
- Safety goggles
1. Salts Dissolving in Water
Example: Ammonium nitrate dissolving. 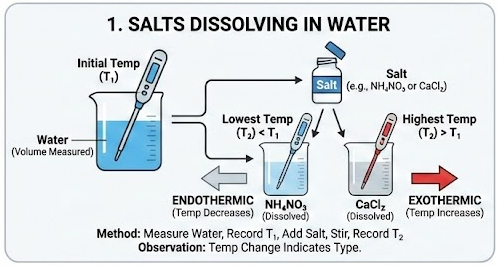
Method:
- Measure a known volume of water into the cup.
- Record the initial temperature.
- Add a known mass of salt.
- Stir until dissolved.
- Record the lowest or highest temperature reached.
Observation:
- Temperature decreases → endothermic (e.g. \( \mathrm{NH_4NO_3} \)).
- Temperature increases → exothermic (e.g. \( \mathrm{CaCl_2} \)).
2. Neutralisation Reactions
Example:
\( \mathrm{HCl(aq) + NaOH(aq) \rightarrow NaCl(aq) + H_2O(l)} \)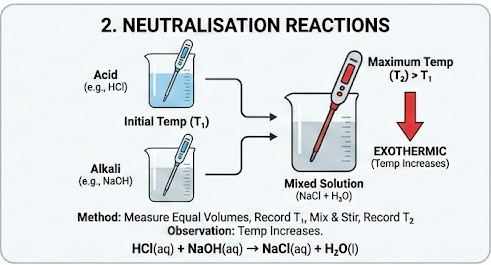
Method:
- Measure equal volumes of acid and alkali.
- Record initial temperature.
- Mix quickly and stir.
- Record maximum temperature.
Observation:
- Temperature increases.
- Reaction is exothermic.
3. Displacement Reactions
Example:
\( \mathrm{Zn(s) + CuSO_4(aq) \rightarrow ZnSO_4(aq) + Cu(s)} \)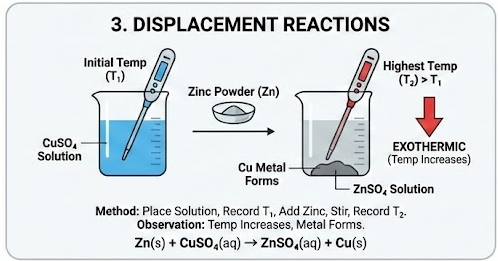
Method:
- Place copper sulfate solution in cup.
- Record initial temperature.
- Add zinc powder.
- Stir and record highest temperature reached.
Observation:
- Temperature increases.
- Copper metal forms.
- Reaction is exothermic.
4. Combustion Reactions
Example: Burning ethanol.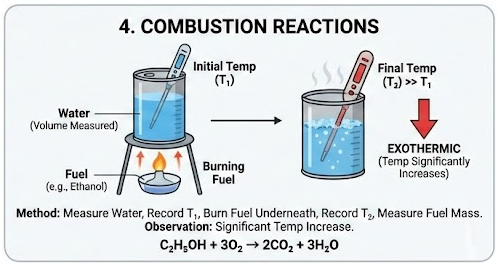
\( \mathrm{C_2H_5OH + 3O_2 \rightarrow 2CO_2 + 3H_2O} \)
Method:
- Measure a known volume of water into a metal can.
- Record initial temperature.
- Burn fuel underneath.
- Record final temperature.
- Measure mass of fuel burned.
Observation:
- Temperature increases significantly.
- Reaction is exothermic.
| Type of Change | Typical Temperature Change | Energy Type |
|---|---|---|
| Salt dissolving | Increase or decrease | Exo or endo |
| Neutralisation | Increase | Exothermic |
| Displacement | Increase | Exothermic |
| Combustion | Increase | Exothermic |
Sources of Error (Exam Focus)
- Heat loss to surroundings.
- Heat absorbed by cup/thermometer.
- Incomplete combustion (for fuels).
- Delay in recording maximum temperature.
Example 1 (Conceptual):
Why does temperature decrease when ammonium nitrate dissolves?
▶️ Answer/Explanation
The process absorbs heat from the surroundings.
It is endothermic.
Example 2 (Application):
During a displacement reaction the temperature rises by \( 10^\circ\mathrm{C} \). What does this show?
▶️ Answer/Explanation
Heat is released.
The reaction is exothermic.
Example 3 (Hard):
Explain fully why combustion experiments often give lower enthalpy values than expected.
▶️ Answer/Explanation
Heat is lost to the surroundings.
Some heat warms the apparatus.
Combustion may be incomplete.
Therefore the measured temperature rise is smaller.
